Editor's Note: This story was originally published in the December 2004 issue of Scientific American.
In an episode of the classic 1950s television comedy The Honeymooners, Brooklyn bus driver Ralph Kramden loudly explains to his wife, Alice, “You know that I know how easy you get the virus.” Half a century ago even regular folks like the Kramdens had some knowledge of viruses—as microscopic bringers of disease. Yet it is almost certain that they did not know exactly what a virus was. They were, and are, not alone.
For about 100 years, the scientific community has repeatedly changed its collective mind over what viruses are. First seen as poisons, then as life-forms, then biological chemicals, viruses today are thought of as being in a gray area between living and nonliving: they cannot replicate on their own but can do so in truly living cells and can also affect the behavior of their hosts profoundly. The categorization of viruses as nonliving during much of the modern era of biological science has had an unintended consequence: it has led most researchers to ignore viruses in the study of evolution. Finally, however, scientists are beginning to appreciate viruses as fundamental players in the history of life.
It is easy to see why viruses have been difficult to pigeonhole. They seem to vary with each lens applied to examine them. The initial interest in viruses stemmed from their association with diseases—the word “virus” has its roots in the Latin term for “poison.” In the late 19th century researchers realized that certain diseases, including rabies and foot-and-mouth, were caused by particles that seemed to behave like bacteria but were much smaller. Because they were clearly biological themselves and could be spread from one victim to another with obvious biological effects, viruses were then thought to be the simplest of all living, gene-bearing life-forms.
On supporting science journalism
If you're enjoying this article, consider supporting our award-winning journalism by subscribing. By purchasing a subscription you are helping to ensure the future of impactful stories about the discoveries and ideas shaping our world today.
Their demotion to inert chemicals came after 1935, when Wendell M. Stanley and his colleagues, at what is now the Rockefeller University in New York City, crystallized a virus— tobacco mosaic virus—for the first time. They saw that it consisted of a package of complex biochemicals. But it lacked essential systems necessary for metabolic functions, the biochemical activity of life. Stanley shared the 1946 Nobel Prize— in chemistry, not in physiology or medicine—for this work.
Further research by Stanley and others established that a virus consists of nucleic acids (DNA or RNA) enclosed in a protein coat that may also shelter viral proteins involved in infection. By that description, a virus seems more like a chemistry set than an organism. But when a virus enters a cell (called a host after infection), it is far from inactive. It sheds its coat, bares its genes and induces the cell’s own replication machinery to reproduce the intruder’s DNA or RNA and manufacture more viral protein based on the instructions in the viral nucleic acid. The newly created viral bits assemble and, voilà, more virus arises, which also may infect other cells.
These behaviors are what led many to think of viruses as existing at the border between chemistry and life. More poetically, virologists Marc H. V. van Regenmortel of the University of Strasbourg in France and Brian W. J. Mahy of the Centers for Disease Control and Prevention have recently said that with their dependence on host cells, viruses lead “a kind of borrowed life.” Interestingly, even though biologists long favored the view that viruses were mere boxes of chemicals, they took advantage of viral activity in host cells to determine how nucleic acids code for proteins: indeed, modern molecular biology rests on a foundation of information gained through viruses.
Molecular biologists went on to crystallize most of the essential components of cells and are today accustomed to thinking about cellular constituents—for example, ribosomes, mitochondria, membranes, DNA and proteins—as either chemical machinery or the stuff that the machinery uses or produces. This exposure to multiple complex chemical structures that carry out the processes of life is probably a reason that most molecular biologists do not spend a lot of time puzzling over whether viruses are alive. For them, that exercise might seem equivalent to pondering whether those individual subcellular constituents are alive on their own. This myopic view allows them to see only how viruses co-opt cells or cause disease. The more sweeping question of viral contributions to the history of life on earth, which I will address shortly, remains for the most part unanswered and even unasked.
To Be or Not to Be
The seemingly simple question of whether or not viruses are alive, which my students often ask, has probably defied a simple answer all these years because it raises a fundamental issue: What exactly defines “life?” A precise scientific definition of life is an elusive thing, but most observers would agree that life includes certain qualities in addition to an ability to replicate. For example, a living entity is in a state bounded by birth and death. Living organisms also are thought to require a degree of biochemical autonomy, carrying on the metabolic activities that produce the molecules and energy needed to sustain the organism. This level of autonomy is essential to most definitions.
Viruses, however, parasitize essentially all biomolecular aspects of life. That is, they depend on the host cell for the raw materials and energy necessary for nucleic acid synthesis, protein synthesis, processing and transport, and all other biochemical activities that allow the virus to multiply and spread. One might then conclude that even though these processes come under viral direction, viruses are simply nonliving parasites of living metabolic systems. But a spectrum may exist between what is certainly alive and what is not.
A rock is not alive. A metabolically active sack, devoid of genetic material and the potential for propagation, is also not alive. A bacterium, though, is alive. Although it is a single cell, it can generate energy and the molecules needed to sustain itself, and it can reproduce. But what about a seed? A seed might not be considered alive. Yet it has a potential for life, and it may be destroyed. In this regard, viruses resemble seeds more than they do live cells. They have a certain potential, which can be snuffed out, but they do not attain the more autonomous state of life.
Another way to think about life is as an emergent property of a collection of certain nonliving things. Both life and consciousness are examples of emergent complex systems. They each require a critical level of complexity or interaction to achieve their respective states. A neuron by itself, or even in a network of nerves, is not conscious—whole brain complexity is needed. Yet even an intact human brain can be biologically alive but incapable of consciousness, or “brain-dead.” Similarly, neither cellular nor viral individual genes or proteins are by themselves alive. The enucleated cell is akin to the state of being braindead, in that it lacks a full critical complexity. A virus, too, fails to reach a critical complexity. So life itself is an emergent, complex state, but it is made from the same fundamental, physical building blocks that constitute a virus. Approached from this perspective, viruses, though not fully alive, may be thought of as being more than inert matter: they verge on life.
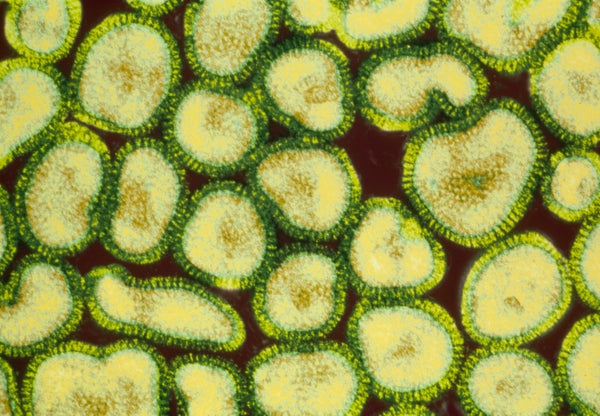
In fact, in October, French researchers announced findings that illustrate afresh just how close some viruses might come. Didier Raoult and his colleagues at the University of the Mediterranean in Marseille announced that they had sequenced the genome of the largest known virus, Mimivirus, which was discovered in 1992. The virus, about the same size as a small bacterium, infects amoebae. Sequence analysis of the virus revealed numerous genes previously thought to exist only in cellular organisms. Some of these genes are involved in making the proteins encoded by the viral DNA and may make it easier for Mimivirus to co-opt host cell replication systems. As the research team noted in its report in the journal Science, the enormous complexity of the Mimivirus’s genetic complement “challenges the established frontier between viruses and parasitic cellular organisms.”
Impact on Evolution
Debates over whether to label viruses as living lead naturally to another question: Is pondering the status of viruses as living or nonliving more than a philosophical exercise, the basis of a lively and heated rhetorical debate but with little real consequence? I think the issue is important, because how scientists regard this question infl uences their thinking about the mechanisms of evolution.
Viruses have their own, ancient evolutionary history, dating to the very origin of cellular life. For example, some viral- repair enzymes—which excise and resynthesize damaged DNA, mend oxygen radical damage, and so on— are unique to certain viruses and have existed almost unchanged probably for billions of years.
Nevertheless, most evolutionary biologists hold that because viruses are not alive, they are unworthy of serious consideration when trying to understand evolution. They also look on viruses as coming from host genes that somehow escaped the host and acquired a protein coat. In this view, viruses are fugitive host genes that have degenerated into parasites. And with viruses thus dismissed from the web of life, important contributions they may have made to the origin of species and the maintenance of life may go unrecognized. (Indeed, only four of the 1,205 pages of the 2002 volume The Encyclopedia of Evolution are devoted to viruses.)
Of course, evolutionary biologists do not deny that viruses have had some role in evolution. But by viewing viruses as inanimate, these investigators place them in the same category of influences as, say, climate change. Such external infl uences select among individuals having varied, genetically controlled traits; those individuals most able to survive and thrive when faced with these challenges go on to reproduce most successfully and hence spread their genes to future generations.
But viruses directly exchange genetic information with living organisms—that is, within the web of life itself. A possible surprise to most physicians, and perhaps to most evolutionary biologists as well, is that most known viruses are persistent and innocuous, not pathogenic. They take up residence in cells, where they may remain dormant for long periods or take advantage of the cells’ replication apparatus to reproduce at a slow and steady rate. These viruses have developed many clever ways to avoid detection by the host immune system— essentially every step in the immune process can be altered or controlled by various genes found in one virus or another.
Furthermore, a virus genome (the entire complement of DNA or RNA) can permanently colonize its host, adding viral genes to host lineages and ultimately becoming a critical part of the host species’ genome. Viruses therefore surely have effects that are faster and more direct than those of external forces that simply select among more slowly generated, internal genetic variations. The huge population of viruses, combined with their rapid rates of replication and mutation, makes them the world’s leading source of genetic innovation: they constantly “invent” new genes. And unique genes of viral origin may travel, finding their way into other organisms and contributing to evolutionary change.
Data published by the International Human Genome Sequencing Consortium indicate that somewhere between 113 and 223 genes present in bacteria and in the human genome are absent in well-studied organisms—such as the yeast Saccharomyces cerevisiae, the fruit fly Drosophila melanogaster and the nematode Caenorhabditis elegans—that lie in between those two evolutionary extremes. Some researchers thought that these organisms, which arose after bacteria but before vertebrates, simply lost the genes in question at some point in their evolutionary history. Others suggested that these genes had been transferred directly to the human lineage by invading bacteria.
My colleague Victor DeFilippis of the Vaccine and Gene Therapy Institute of the Oregon Health and Science University and I suggested a third alternative: viruses may originate genes, then colonize two different lineages—for example, bacteria and vertebrates. A gene apparently bestowed on humanity by bacteria may have been given to both by a virus.
In fact, along with other researchers, Philip Bell of Macquarie University in Sydney, Australia, and I contend that the cell nucleus itself is of viral origin. The advent of the nucleus— which differentiates eukaryotes (organisms whose cells contain a true nucleus), including humans, from prokaryotes, such as bacteria—cannot be satisfactorily explained solely by the gradual adaptation of prokaryotic cells until they became eukaryotic. Rather the nucleus may have evolved from a persisting large DNA virus that made a permanent home within prokaryotes. Some support for this idea comes from sequence data showing that the gene for a DNA polymerase (a DNAcopying enzyme) in the virus called T4, which infects bacteria, is closely related to other DNA polymerase genes in both eukaryotes and the viruses that infect them. Patrick Forterre of the University of Paris-Sud has also analyzed enzymes responsible for DNA replication and has concluded that the genes for such enzymes in eukaryotes probably have a viral origin.
From single-celled organisms to human populations, viruses affect all life on earth, often determining what will survive. But viruses themselves also evolve. New viruses, such as the AIDS-causing HIV-1, may be the only biological entities that researchers can actually witness come into being, providing a real-time example of evolution in action.
Viruses matter to life. They are the constantly changing boundary between the worlds of biology and biochemistry. As we continue to unravel the genomes of more and more organisms, the contributions from this dynamic and ancient gene pool should become apparent. Nobel laureate Salvador Luria mused about the viral influence on evolution in 1959. “May we not feel,” he wrote, “that in the virus, in their merging with the cellular genome and reemerging from them, we observe the units and process which, in the course of evolution, have created the successful genetic patterns that underlie all living cells?” Regardless of whether or not we consider viruses to be alive, it is time to acknowledge and study them in their natural context—within the web of life.