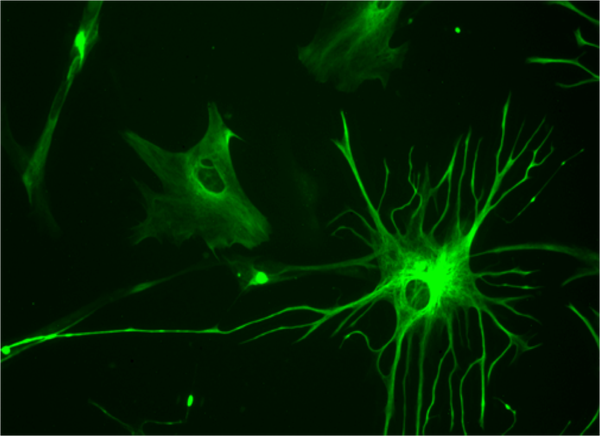
Star-shaped cells called astrocytes—often characterized as “helper” cells—may contribute to damage caused by brain injury and disease by turning toxic and destroying neurons, according to study results published Wednesday in Nature.
Astrocytes are one of the three types of glial, or non-neuronal, cells, the most abundant kind found in the brain. They are widely regarded as support cells that nourish neurons and pack the spaces between them, but it is becoming increasingly clear that they play other important roles in normal, healthy brain function. They can synthesize neurotransmitters to send signals among glial cells, and form networks that regulate neuronal activity.
Astrocytes can also react to brain injury and disease in various ways. Following nerve damage, for example, they form scar tissue that can aid in the regeneration of severed fibers. But they are also implicated in a wide variety of neurological and psychiatric diseases.
On supporting science journalism
If you're enjoying this article, consider supporting our award-winning journalism by subscribing. By purchasing a subscription you are helping to ensure the future of impactful stories about the discoveries and ideas shaping our world today.
The new findings show that under certain conditions astrocytes can transform into a highly toxic state and kill other types of brain cells. But by providing fresh insights into the process of neurodegeneration, they could eventually lead to novel treatments for a wide range of diseases.
The results of the new study build on earlier work from the laboratory of neurobiologist Ben Barres at Stanford University, which isolated reactive astrocytes from mice with experimentally induced stroke and brain inflammation, and used DNA chip technology to create genetic profiles of the cells. Those studies found that stroke and inflammation triggered the conversion of astrocytes into two reactive cell types: A1 cells, which ramp up their production of immune system molecules called complement proteins, and A2 cells, which express high levels of growth factor proteins that promote the survival of brain cells and formation of connections between them.
Complement proteins attach themselves to brain synapses, “tagging” them for destruction by microglia—the brain's resident immune cells—leading the researchers to hypothesize that A1 astrocytes may be harmful.
In the Nature study, led by Stanford neuroscientist Shane Liddelow, a postdoctoral fellow in Barres's lab, the researchers induced brain inflammation in normal, healthy mice as well as in genetically engineered animals lacking microglial cells. They then examined the astrocytes in both groups of animals. In the normal mice inflammation prompted the formation of A1 astrocytes. No such response was seen in the genetically engineered animals, suggesting microglial cells induce the development of these astrocytes.
To test this idea further they grew microglia in Petri dishes, treated them with a chemical that activates an inflammatory response, and then transferred the growth medium into another Petri dish containing astrocytes. This induced the formation of A1 astrocytes whereas the growth medium from nonactivated microglia did not.
Further experiments revealed activated microglia secrete a cocktail of three immune-system chemicals called cytokines—interleukin-1 alpha (Il-1α), tumor necrosis factor (TNF) and complement component 1, q subcomponent (C1q). Addition of all three molecules to astrocyte cultures generated A1 astrocytes, but antibodies that neutralize any one of them blocked this effect. Confirming this, the researchers also found that three different strains of genetically engineered mice—each missing one of the cytokine genes—all had a significantly reduced ability to generate A1 astrocytes.
In a healthy brain astrocytes form networks that are vital for the formation of connections—synapses—between neurons, and they also regulate signals that are transmitted across those synapses. In injury and disease situations, however, they can contribute to brain repair by engulfing and clearing away dead cells and other cellular debris via a process called phagocytosis.
To determine whether A1 astrocytes retain either the building or repair functions, Liddelow and his colleagues grew retinal ganglion cells, which form the optic nerve, together with either A1 astrocytes or nonactivated astrocytes. Petri dishes with A1 astrocytes added to them contained approximately 50 percent fewer synaptic connections compared with those holding nonactivated ones. And the remaining synapses were significantly weaker, producing smaller and less frequent signals than those in the dishes containing nonactivated astrocytes.
The researchers also found high concentrations of A1 astrocyte growth medium killed various types of neurons as well as another type of glial cells, oligodendrocytes, which synthesize the insulating sheathing, or myelin, on neurons’ long, wirelike fibers. A1 astrocytes also have a markedly reduced ability to engulf cellular debris compared with nonactivated ones.
When injected into mice with crushed optic nerves, rather than protect them A1 astrocytes killed the retinal ganglion cells with damaged fibers. This was prevented by injection of the anti-cytokine antibodies, however, and did not occur in mutant mice lacking two or three of the cytokine genes.
So it seems that astrocytes and microglia are partners in crime. Microglia make up a surveillance system that monitors the state of brain tissue, mounting the appropriate response to injury. The new results show they use cytokines to signal astrocytes and transform them from a state that promotes the survival of neurons to one that is highly toxic. “These are very interesting and exciting observations,” says molecular physiologist Frank Kirchoff of the University of Saarland in Germany, who was not involved in the new research. “Astrocytes are normally regarded as the good guys, but here we have convincing evidence of a well-defined mechanism for how they contribute to different types of pathologies.”
To find out if this process might contribute to neurodegeneration, Liddelow and his colleagues examined postmortem brain tissue from patients with Alzheimer’s, Huntington’s and Parkinson’s diseases as well as multiple sclerosis and amyotrophic lateral sclerosis. All the tissue samples they examined contained large numbers of A1 astrocytes, suggesting the cells may drive neurodegeneration. This insight might potentially point to a new path for treatment of these diseases. “We cannot simply target all astrocytes because such a blunt-force approach would also remove the nonreactive ones,” Liddelow says. “The results suggest a regulated and specific astrocyte response that is likely to depend on time and context, so we must be careful to develop drugs that combat only the specific type of reactivity.”
Liddelow and his colleagues are trying to identify the toxic signal that A1 astrocytes release. “That will provide a wealth of information about why neurons die in neurodegenerative diseases such as Alzheimer’s, and why there is demyelination of axons in diseases such as multiple sclerosis.” Finding some means of regulating that signal might hint at ideas for treating these diseases.