The attacks began in her early 20s; for two years now Stephanie* has been living under the pall of migraine. It usually starts with a visual disturbance called an aura—shimmering zigzag lines that move across her field of vision and gradually expand into blackness, blotting out her sight. Then comes pounding pain mainly on the left side of her head. Adding to her misery is an exquisite sensitivity to light, sound and smell that makes ordinary stimuli—even perfume—unbearable and the headache even worse.
When she arrived at the neurology clinic in Halifax, Nova Scotia, where one of us (Purdy) practices, she said she had tried numerous medications, but all had either failed or triggered intolerable side effects. Meanwhile, over the past year, her symptoms have become more extreme and frightening. Now as her vision blurs, a tingling sensation slowly moves upward from her right hand through her arm and sometimes into her face and tongue—a sign that blinding head pain is about to strike. At the same time, she begins to have trouble finding her words and making herself intelligible to others. She worries that migraine will lead to a stroke, which can be a genuine risk.
For some people, migraine is an occasional bother; for others, a persistent scourge. For people such as Stephanie, the attacks can involve bizarre alterations in perception and sensation. But in its various forms, migraine is one of the most common of neurological conditions, affecting an estimated 39 million people in the U.S.—about one in five women and one in 16 men.
On supporting science journalism
If you're enjoying this article, consider supporting our award-winning journalism by subscribing. By purchasing a subscription you are helping to ensure the future of impactful stories about the discoveries and ideas shaping our world today.
It is a malady that often defies treatment and prevention. According to population studies in the U.S., only one quarter of those with episodic migraine (fewer than 15 days of headache a month) and fewer than 5 percent of those with chronic migraine (15 or more days a month) have seen a health care provider and received an accurate diagnosis and appropriate therapy. The newest class of drugs on the market dates back to the early 1990s. Taken after an attack has started, these medications resolve the pain in fewer than a third of those who use them, and they have potential side effects that rule them out for many.
Now, though, people with migraine have reason to hope that their misery may soon be behind them. Breakthroughs in understanding brain networks and the chemical messengers that cause migraine symptoms have spawned a number of sophisticated new treatments that halt attacks or prevent them from beginning. And just as new insights into the brain are generating novel ways to attack the disorder, the study of migraine is revealing some of the brain's secrets.
An Ancient Affliction
Migraine is rivaled only by epilepsy for the title of oldest-known neurological disorder. The Egyptians described it in medical documents in 1200 B.C., although credit for its discovery as a distinct condition usually goes to Aretaeus of Cappadocia, whose second-century writings gave accounts of individuals with repeated attacks of severe, one-sided headaches and vomiting.
The severity of migraine varies widely. The lucky might have a very bad headache a few times a year, treat it with an over-the-counter painkiller and never bother to seek a doctor's advice. For most, however, attacks occur once or twice a month, and for about a quarter of U.S. migraine sufferers, the attacks become more frequent, with symptoms eventually occurring daily or nearly so. For them, while the disease is not in itself life-threatening, it certainly is life-altering.

For some people migraine begins with a shimmering visual aura that can partially blot out vision. The effect is simulated here. Credit: Andrew Too Boon Tan Getty Images (bike in front of store); Scientific American Mind (blur)
Although the most debilitating element of migraine is the pain, the most distinctive (and fascinating for neurologists like us) is the aura, which affects about a third of migraineurs. Most often the aura is a visual phenomenon consisting of jagged lines that produce an area of blurry or reduced vision, sometimes with a blind spot at the center. Imaging studies have revealed that this so-called classical aura originates in the visual cortex at the back of the brain and spreads forward over the course of several minutes—a phenomenon that is known as cortical spreading depression [see “The Long, Slow Wave” at bottom].
Most of those who have no visual aura will still experience premonitory symptoms—yawning, fatigue, mood changes, neck pain, sensitivity to light—that may serve as a warning of an imminent headache. Migraine is also linked to risks of other maladies, such as ischemic stroke, depression and epilepsy. The risk of stroke is especially high in women, particularly those who smoke or take medications containing estrogen, such as birth-control pills.
Clinicians have long observed that migraine tends to run in families. Recent research has identified more than three dozen genes that appear to be associated with the disorder. These include genes responsible for so-called channels and transporters that sit on the surface of neurons and other brain cells. These structures control the traffic of ions (such as sodium, potassium and calcium) in and out of the cells and neurotransmitters across the synapses—and thus the excitability of certain brain cells and brain networks. Other genes associated with migraine are responsible for generating pain and maintaining the health of blood vessels. Taken together, these genetic variations may explain the hyperexcitability of the brain and the extreme sensitivity to light, sound and odors, the pain, and the vascular disorders, including stroke, associated with migraine.
The Quest for Relief
The earliest treatments for migraine were rooted in superstition and witchcraft, ranging from bloodletting to opening a hole in the skull to release evil spirits. In the 19th century, migraine was considered to be a psychosomatic illness, along with other afflictions suffered principally by women. That notion began to change in the middle of the 20th century. A series of elegant experiments in the 1940s by New York Hospital–Cornell Medical Center neurologist Harold Wolff led to the modern vascular theory of migraine—notably, that the pain of migraine is from dilation and distention of blood vessels outside and inside the skull. Wolff measured the amplitude of pulsations of blood vessels in the scalp during migraine attacks and after the administration of a drug that constricted the vessels. His findings led to the adoption of the first true migraine medicine: ergotamine tartrate, a powerful vasoconstrictor derived from the ergot fungus; it brought pain relief that coincided with artery constriction in the scalp.
Then, in the 1970s and 1980s, investigators at the Erasmus University Rotterdam in the Netherlands and the University of New South Wales in Sydney, Australia, noted an association between migraine and serotonin: levels of the neurotransmitter went down in the blood and up in the urine during attacks. In other words, the body was losing serotonin. They also found that administering serotonin to people during an attack relieved pain, just as ergotamine did. The thinking at the time was that loss of serotonin caused blood vessels to lose tone and dilate, bringing on migraine pain.
As treatments, though, ergotamine and serotonin have serious problems. Both have troubling side effects, including nausea, vomiting and cramping—which can already be problems for migraine sufferers. Ergotamine can also cause dangerous reductions in blood flow.
In the 1970s Patrick Humphrey, a pharmacologist then at British pharmaceutical company Glaxo, began to look for a way to reproduce the beneficial effects of serotonin without the harmful ones. Humphrey was working under the assumption that the dilation of the blood vessels inside and outside the skull was responsible for migraine headaches and that drugs that could bind to serotonin receptors might provide relief. So he set out to design a drug that did just that. The result, after a decade of work, was sumatriptan, which, like ergotamine, both eased pain and constricted blood vessels. It would be the first in the triptan family of drugs.
Triptans, too, have their limitations. They bring complete pain relief to only about 30 percent of migraine sufferers, and for many of them, the headache returns the same day. Triptans may also trigger a number of unpleasant side effects, including sleepiness, dizziness, tingling, tightness in the chest, and a reddish flushing of the face and neck. And because these drugs can constrict blood vessels throughout the body, people with heart disease or a history of stroke cannot use them.
But the triptans were a major breakthrough for millions of sufferers who could now take a pill or injection and stop a debilitating headache within 30 minutes. They were also a triumph in the science of drug design—except that the understanding of the underlying neurobiology was not altogether right. As further research would soon reveal, the primary cause of migraine pain is not dilation of blood vessels in the head, and the primary factor by which triptans help is not by constricting those vessels. Something else was going on, and the design of better drugs would depend on figuring out what that was.
Guided Missiles for Migraine
In the 1980s, at around the same time that Humphrey was working on triptans, neuroscientist Lars Edvinsson of Lund University in Sweden found a compound known as calcitonin gene–related peptide (CGRP) in the nerves that surround the blood vessels inside the skull. The compound had only recently been discovered in the central and peripheral nervous system, and evidence suggested that it served as a chemical messenger of pain. CGRP is also a potent vasodilator, and Edvinsson, a specialist in the brain's blood supply, hypothesized that it may contribute to the development of migraine headache.
Over the next two decades experiments by numerous investigators confirmed that idea. Researchers found, for instance, that blood levels of CGRP rose during migraine attacks and returned to normal after a dose of sumatriptan relieved the headache. Especially convincing, CGRP consistently triggered a migraine attack when infused into the bloodstream of migraine sufferers. Studies in both animals and humans showed that CGRP and its receptors are found in brain structures such as the hypothalamus and cerebellum, which were long thought to play a role in generating migraine attacks. They are also present in the trigeminal nerve, a key cranial nerve involved in processing sensory signals that was also implicated in migraine. Furthermore, CGRP turns out to be one of the chemical substances released during cortical spreading depression, the putative mechanism of migraine aura.
Click or tap to enlarge

Credit: Tami Tolpa; Source: “CGRP-Receptor Antagonists: A Fresh Approach to Migraine Therapy?” by Paul L. Durham, in New England Journal of Medicine, Vol. 350, No. 11; March 11, 2004
In the early 2000s scientists at pharmaceutical company Boehringer Ingelheim in Germany synthesized a small molecule designed to bind to the receptor for CGRP and block its activity—a category of substances known as receptor antagonists. A study involving 126 patients published in the New England Journal of Medicine in 2004 confirmed that this molecule, given intravenously, halted migraine headaches in some patients, and it did so without constricting blood vessels in the head. This was a crucial discovery: it demonstrated that the long-reigning vascular model of migraine was not wholly correct and that constriction of blood vessels was not essential to bringing relief.
Despite the promising results, progress in developing CGRP receptor antagonists faltered because of a serious side effect: liver toxicity that arose in trials of three of these drugs. Researchers would have to find another way forward.
Some decided to target CGRP or its receptor with a monoclonal antibody. Antibodies are large proteins that can be directed with precision to a single target much like a laser-guided missile. Drug developers typically produce them from clones of a single parent immune cell, which is why they are described as monoclonal. Like other proteins, they are metabolized into amino acids by tissues throughout the body rather than taxing the kidneys or liver. Thus, although they may have side effects associated with blocking their specific target (such as CGRP), they should not cause “off-target” side effects or toxicity such as kidney damage.
Antibodies are too large to pass through the blood-brain barrier, and yet these new drugs have performed impressively in preventing migraines in preliminary studies. One possible mechanism: by blocking CGRP in trigeminal nerve pathways outside the brain, the antibodies may reduce signaling between the peripheral and central nervous systems, decreasing pain signals entering the brain.
Since 2012, numerous researchers—including one of us (Dodick)—have conducted placebo-controlled clinical trials involving a total of more than 10,000 patients. A large proportion of the trial patients have used the treatment for more than a year, without side effects, aside from some redness at the injection site. For up to 70 percent of them, the number of days with headache dropped by more than half, and up to one in six patients became completely migraine-free while the treatment lasted. The improvement appeared as soon as three days after administration of the antibody began. Pharmaceutical company Amgen recently completed its pivotal phase III studies and will soon file for FDA clearance. Three other companies—Alder BioPharmaceuticals, Eli Lilly and Teva Pharmaceutical Industries—are in the midst of their phase III trials. Barring unforeseen issues, the first of these antibodies will become available in late 2017 or early 2018.
The monoclonal antibodies will be a leap forward from currently available preventive treatments, which include beta blockers such as propranolol and a variety of blood pressure medications. The older drugs have similar response rates, but a noticeable effect might take weeks or months to appear. Their dose often needs to be increased over time, and the side effects, such as weight gain, hair loss, cognitive dysfunction and sedation, may deter patients from reaching an effective dose or continuing with the drug at all. In fact, more than 85 percent of patients who start treatment with today's preventive drugs stop taking them within a year.
Still, monoclonal antibody treatment will not be for everyone. Patients will need to take them either intravenously, requiring a trip to the doctor every three months, or via a monthly self-administered injection. And questions remain over the long-term safety of blocking a protein found throughout the body. This concern is particularly relevant to patients with cardiovascular disease and hypertension because CGRP is thought to be important in maintaining the tone of blood vessels and compensating for low blood supply to the brain and heart during strokes and heart attacks. Nor is it at all clear that the drugs would be safe for use during pregnancy—an important consideration because most migraineurs seen in clinical practice are women of child-bearing age. (Migraines often abate at menopause.)
For patients who cannot use the antibodies or are not adequately helped by them and for the many who would prefer to treat migraines only when they arise, there remains a need for safe and effective on-demand treatment for acute attacks. Ideally such pain relievers would be in pill form and would not constrict blood vessels, as triptans do. Two classes of medication now under development show promise on all these counts. Clinical trials of newer CGRP receptor antagonists have demonstrated them to be about as effective as triptans but without the toxic effects. The liver toxicity seen in earlier trials appears to have been specific to the drugs and not to blocking CGRP.
And a new family of drugs called ditans, which target serotonin receptors, is showing great promise. Lasmiditan, under development by CoLucid Pharmaceuticals, is selective for serotonin receptors located only on neurons and not blood vessels, which means it should be safe for the 20 percent of migraine patients who have cardiovascular risk factors. In a recent clinical trial of lasmiditan by CoLucid involving 2,231 subjects who received treatment for a single migraine, a third of patients were pain-free within two hours. (Dodick helped to design the trial and analyze the results.) This success rate is similar to that of triptans but without the risks associated with constriction of blood vessels. In fact, more than 80 percent of the patients in the study had one or more cardiovascular risk factors, and no safety issues arose.
Hope on the Horizon
The new treatments making their way to market reflect tremendous strides in identifying the sites in the brain where migraine arises and the mechanisms that generate it. The result is medication with greater specificity and fewer side effects than older remedies. The creation of a successful drug often leads to a virtuous cycle of further insights into disease mechanisms and the next generation of treatments. This was true of the triptans and will almost certainly be true for the drugs in development today. The mere fact that a large molecule such as an antibody with no likely access to the brain can prevent attacks originating there is already transforming how we think not only about migraine but the very way in which the brain works.
Most important, patients such as Stephanie who have been unable to find relief with current meds will—if all goes well—finally get the attention and treatment they deserve. Stephanie has tried at least 10 medications to control her migraines, as well as numerous alternative therapies, from acupuncture to yoga to special diets—without effect. As someone whose symptoms place her at risk of a stroke, she was excited to hear that potentially safer drugs are on the horizon. “That sounds wonderful,” she said. “I can't wait!” As clinicians who see far too many patients whose migraines resist current treatments, we feel the same way.
*The patient’s name has been changed, along with a few details of her case, to protect her privacy.
The Long, Slow Wave
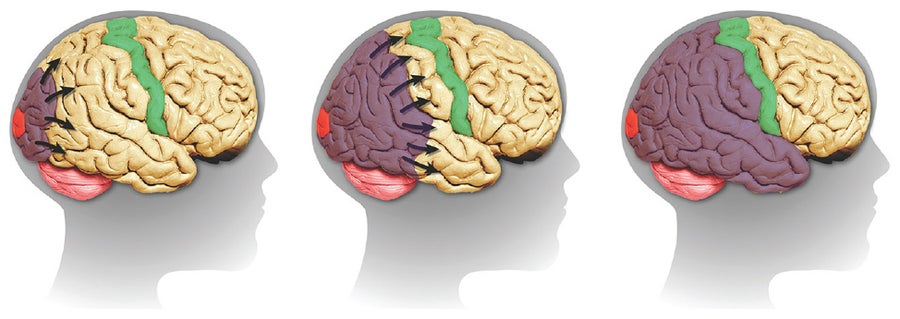
Starting in the visual cortex at the back of the brain (left), cortical spreading depression moves forward as a wave of electrical activity (purple) at about two to three millimeters a minute, setting off a visual aura. When it reaches the parietal and temporal cortices (center), speech difficulties may ensue, and then, at the sensory strip (right, green), tingling sensations may occur in a limb or the head. Credit: Courtesy of Richard Hargreaves
One of the enduring mysteries of migraine is the relation between the splitting headache and the strange sensory symptoms that sometimes precede it. A phenomenon known as cortical spreading depression, or CSD, may be responsible for both. CSD is characterized by a wave of electrical activity that spreads slowly over the cortex, followed by a wave of quietude, producing symptoms that are related to the part of the brain through which the wave is traveling.
After any of a number of factors set off an attack—stress, bright lights, hormone changes, lack of sleep—CSD typically starts in the visual cortex in the back of the brain, triggering the zigzag patterns and blurred vision characteristic of classic aura. The wave then may travel to the sensory strip in the parietal lobe, inducing a marching disturbance from the hand up to the face and tongue. For some sufferers, speech is stricken next, bringing on aphasia.
As it traverses the brain, CSD can also stimulate pain-detecting neurons either directly or through inflammation that excites fibers supplying the sensitive outer surface of the brain. These pain fibers subsequently release a variety of chemical or protein neurotransmitters—among them the peptide CGRP—that are capable of transmitting pain signals from the peripheral to the central nervous system.
Two thirds of people with migraine, however, do not have an aura. For them, the migraine trigger remains an active area of investigation. CSD might occur in cortical or subcortical brain tissue without giving rise to sensory symptoms. Or different mechanisms might generate an attack in subcortical brain structures that help to process light, sound and other sensory stimuli and influence pain-sensing neurons in the brain. Or both types of mechanisms might work together. In either case, CSD is an elegant bridge between migraine pain and its remarkable neurological symptoms. —R.A.P. and D.W.D.

