Oh dear—π day 2016 has gone by and I still haven’t memorized p in what I used to call the ‘‘elementary’’ way that I invented some decades ago. Somebody recently suggested the much better name ‘‘Chemical π.’’ What can I do to make sure I do it by next π day? I know— I’ll write an article called ‘‘Chemical π’’ for The Mathematical Intelligencer. Here goes:
Let me tell you how Chemical π starts. First, I should remind you that it was proposed by several chemists, including Andreas von Antropoff and Charles Janet, that what is now called the neutron could be regarded as an atom of the element of atomic number 0, which has been called Neutronium and assigned the chemical symbol Nn.
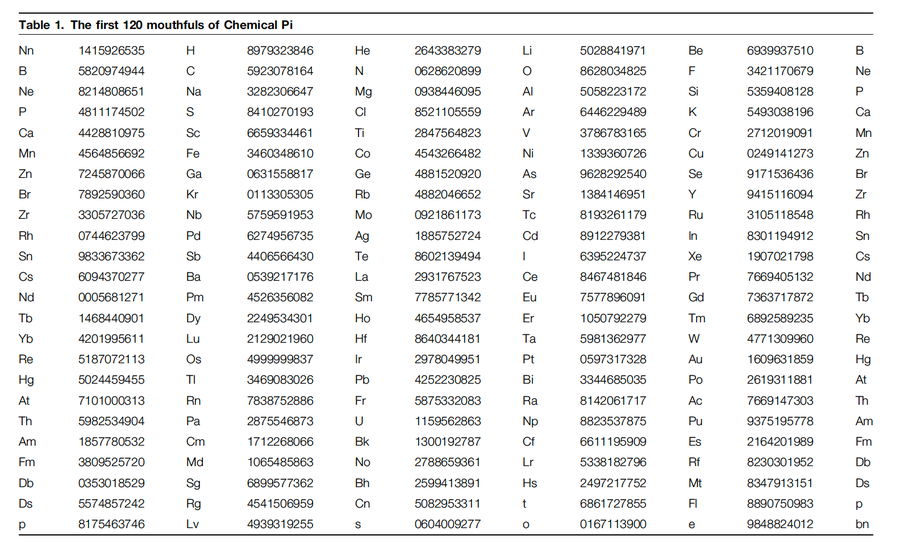
Here is chemical π:
On supporting science journalism
If you're enjoying this article, consider supporting our award-winning journalism by subscribing. By purchasing a subscription you are helping to ensure the future of impactful stories about the discoveries and ideas shaping our world today.
3 Neutronium 1415926535 Hydrogen 8979323846 Helium 2643383279 Lithium 5028841971 Beryllium ...
You get the idea? In between the elements of atomic numbers n and n + 1, we insert the 10 decimal digits of π numbered 10n + 1 to 10n + 10. So for instance, since Lithium and Beryllium have atomic numbers 3 and 4, the 31st to 40th digits of π appear between Li and Be making the ‘‘mouthful’’
Li 5028841971 Be.
Chemical π is a way to learn π with lots of advantages.
It helps to solve the forgetting problem.
If you just remember π as a long string of digits, then your only name for any substring of digits in π is that substring itself. So if you forget that substring, you’ve forgotten what you’ve forgotten! With the chemicals inserted like beads, you can say for example, I’ve forgotten the part between Magnesium and Aluminum and clear this up by quickly looking for it in Table 1 of this paper: Mg 0938446095 Al.
It makes the remembering problem easier.
Mnemo-technicians know that memorization becomes easier if one attaches ‘‘colorful’’ things to the items one is memorizing. In Chemical π, the chemicals are the color.
It divides the digits into easily digested ‘‘mouthfuls,’’ each consisting of two adjacent elements separated by 10 digits of pi.
This gives you a nice trick. Ask a friend to give you any number less than 1200 and then recite π starting at that decimal digit. For instance, if he says 314, you read the gallium (no. 31) mouthful from its fourth digit 1558817 then 48815…
It solves the stopping problem.
When I first learned π to 1000 places, I triumphantly finished ‘‘...1989’’ to my friend John McKay. He said: ‘‘What comes next?’’ I realized he had a point. It was odd to know everything about the first thousand decimal digits but nothing of the next ones. I decided to add an extra 10% as ‘‘play,’’ making 1100 digits and then 10 more digits as ‘‘play in the play,’’ making 1110 and another digit as ‘‘play in the play in the play’’ and then for the play in that I asked him to look up the next digit, not tell me what it was but just whether it was 5 or more so that I would know whether to round up or down. This led to 1111 digits and a bit (binary digit). With the chemical method, the rule is that a chemically pious person need only memorize digits 10n + 1 to 10n + 10 of π when IUPAC (the International Union of Pure and Applied Chemistry) has accepted a final name for the element of atomic number n. Happily this goes just a little bit further than the play-in-the-play-in-the-play method.
What I call the IUPAC ‘‘cipher’’ provides provisional names ending in ium for the elements from 100 to 999 by ‘‘enciphering’’ the digits
0, 1, 2, 3, 4, 5, 6, 7, 8, 9 as nil, un, bi, tri, quad, pent, hex, sept, oct, enn.
In this paper we abbreviate the cipher by omitting any initial letters U or u.
By 2010 IUPAC had accepted permanent names for the elements up to 112 (Copernicum) and a year later it recognized the names Flerovium (114) and Livermorium (116). On January 4 2016, IUPAC accepted that elements 113, 115, 117, and 118 have been constructed, but the institutions that constructed them still don’t seem to have chosen permanent names.

If Charles Janet was right in his prediction that the element with atomic number 120, provisionally Unbinilium, would be the last such element, many people competing in Pi Day celebrations could achieve chemical piety forever!
The nerds who read this paper will probably be interested in Janet’s ‘‘Left Step’’ version of the periodic table, even though it has no connection with the number π. I’ll describe my slight improvement of this table, which is printed in Table 3 with my mnemonic for the chemical elements it contains. It consists of two periods of length 2 = 2 x 12, two of length 8 = 2 x 22, two of length 18 = 2 x 32, and finishes with two of length 32 = 2 x 42. If started with hydrogen as Janet did, this takes us to element number 120, which he thought would be the last element to be constructed. However, this puts Helium in column 1 from the right and the other noble gasses in column 3 (again the right), and has other similar defects. These defects are cured if we start from Neutronium, when in particular the noble gasses form the column 2. This makes element 119 dislodge element 120 from the last place, which might suggest that 119 should be the last to be constructed. However, if element 120 is constructed, then it could be named Jt after Janet and the last mouthful would be Jt 8583616035 bu.
Is it perhaps not too much to hope that when celebrations start at 1.59 on future Pi Days, the pious might spare a thought for the modest inventor of Chemical Pi?
The chemical symbols of the following elements refer to alternative, older names. Sodium = Na = Natrium, Potassium = K = Kalium, Copper = Cu = Cuprum, Silver = Ag = Argentum, Tin = Sn = Stannum, Antimony = Sb = Stibnum, Tungsten = W = Wolfram, Gold = Au = Aurum, Mercury = Hg = Hydrargyrum, Lead = Pb = Plumbum.

