Key concepts
Biology
Biochemistry
Enzymes
Physiology
Chemistry
Introduction
Have you ever wondered how all the food that you eat gets digested? It is not only the acid in your stomach that breaks down your food—many little molecules in your body, called enzymes, help with that too. Enzymes are special types of proteins that speed up chemical reactions, such as the digestion of food in your stomach. In fact, there are thousands of different enzymes in your body that work around-the-clock to keep you healthy and active. In this science activity you will investigate one of these enzymes, called catalase, to find out how it helps to protect your body from damage.
Background
Enzymes are essential for our survival. These proteins, made by our cells, help transform chemicals in our body, functioning as a catalyst. A catalyst gets reactions started and makes them happen faster, by increasing the rate of a reaction that otherwise might not happen at all, or would take too long to sustain life. However, a catalyst does not take part in the reaction itself—so how does this work? Each chemical reaction needs a minimum amount of energy to make it happen. This energy is called the activation energy. The lower the activation energy of a reaction, the faster it takes place. If the activation energy is too high, the reaction does not occur.
On supporting science journalism
If you're enjoying this article, consider supporting our award-winning journalism by subscribing. By purchasing a subscription you are helping to ensure the future of impactful stories about the discoveries and ideas shaping our world today.
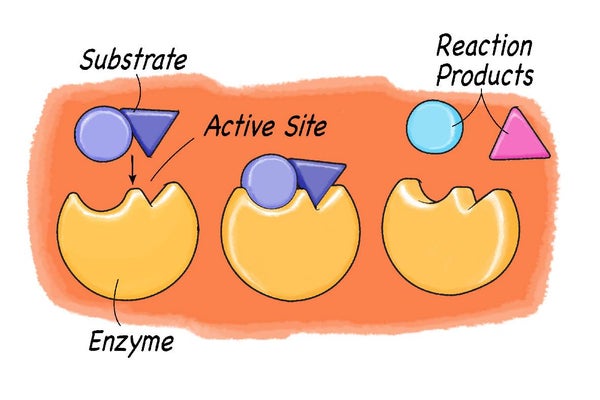
Enzymes have the ability to lower the activation energy of a chemical reaction by interacting with its reactants (the chemicals doing the reacting). Each enzyme has an active site, which is where the reaction takes place. These sites are like special pockets that are able to bind a chemical molecule. The compounds or molecules the enzyme reacts with are called their substrates. The enzyme pocket has a special shape so that only one specific substrate is able to bind to it, just like only one key fits into a specific lock. Once the molecule is bound to the enzyme, the chemical reaction takes place. Then, the reaction products are released from the pocket, and the enzyme is ready to start all over again with another substrate molecule.
Catalase is a very common enzyme that is present in almost all organisms that are exposed to oxygen. The purpose of catalase in living cells is to protect them from oxidative damage, which can occur when cells or other molecules in the body come into contact with oxidative compounds. This damage is a natural result of reactions happening inside your cells. The reactions can include by-products such as hydrogen peroxide, which can be harmful to the body, just as how a by-product of a nice bonfire can be unwanted smoke that makes you cough or stings your eyes. To prevent such damage, the catalase enzyme helps getting rid of these compounds by breaking up hydrogen peroxide (H2O2) into harmless water and oxygen. Do you want to see the catalyze enzyme in action? In this activity you will disarm hydrogen peroxide with the help of catalase from yeast.
Materials
Safety goggles or protective glasses
Five teaspoons of dish soap
One package of dry yeast
Hydrogen peroxide, 3 percent (at least 100 mL)
Three tablespoons
One teaspoon
Five 16-ounce disposable plastic cups
Tap water
Measuring cup
Permanent marker
Paper towel
Workspace that can get wet (and won't be damaged by any spilled hydrogen peroxide or food-colored water)
Food coloring (optional)
Preparation
Take one cup and dissolve the dry yeast in about one-half cup of warm tap water. The water shouldn't be too hot but close to body temperature (37 Celsius). Let the dissolved yeast rest for at least five minutes.
Use the permanent marker to label the remaining four cups from one to four.
To all the labeled cups, add one teaspoon of dish soap.
To cup one no further additions are made at this point.
Before using the hydrogen peroxide, put on your safety goggles to protect your eyes. In case you spill hydrogen peroxide, clean it up with a wet paper towel. If you get it on your skin, make sure to rinse the affected area with plenty of water.
To cup two, add one tablespoon of 3 percent hydrogen peroxide solution. Use a fresh spoon for the hydrogen peroxide.
To cup three, add two tablespoons of the hydrogen peroxide.
To cup four, add three tablespoons of the hydrogen peroxide.
Optionally, you can add a drop of food color to each of the labeled cups. (You can choose a different color for each one for easy identification)
Procedure
Take cup number one and place it in front of you on the work area. With a fresh tablespoon, add one tablespoon of the dissolved yeast solution to the cup and swirl it slightly. What happens after you add the yeast? Do you see a reaction happening?
Place cup number two in front of you and again add one tablespoon of yeast solution to the cup. Once you add the enzyme,does the catalase react with the hydrogen peroxide? Can you see the reaction products being formed?
Add one tablespoon of yeast solution to cup number three. Do you see the same reaction taking place? Is the result different or the same compared to cup number two?
Finally, add one tablespoon of yeast solution to cup number four. Do you see more or less reaction products compared to your previous results? Can you explain the difference?
Place all four cups next to each other in front of you and observe your results. Did the enzymatic reaction take place in all of the cups or was there an exception? How do the results in each cup look different? Why do you think this is the case?
Now, take cup number one and add one additional tablespoon of 3 percent hydrogen peroxide to the cup. Swirl the cup slightly to mix the solution. What happens now? Looking at all your results, what do you think is the limiting factor for the catalase reaction in your cups?
Extra: Repeat this activity, but this time do not add dish soap to all of the reactions. What is different once you remove the dish soap? Do you still see foam formation?
Extra: So far you have observed the effect of substrate (H2O2) concentration on the catalase reaction. What happens if you keep the substrate concentration constant but change the concentration of the enzyme? Try adding different amounts of yeast solution to three tablespoons of hydrogen peroxide, starting with one teaspoon. Do you observe any differences, or does the concentration of catalase not matter in your reaction?
Extra: What happens if the environmental conditions for the enzyme are changed? Repeat the catalase reaction but this time vary conditions such as the pH by adding vinegar (an acid) or baking soda (a base), or change the reaction temperature by heating the solution in the microwave. Can you identify which conditions are optimal for the catalase reaction? Are there any conditions that eliminate the catalase activity?
Extra: Can you find other sources of catalase enzyme that you could use in this activity? Research what other organisms, plants or cells contain catalase and try using these for your reaction. Do they work as well as yeast?
Observations and results
You probably saw lots of bubbles and foam in this activity. What made the foam appear? When the enzyme catalase comes into contact with its substrate, hydrogen peroxide, it starts breaking it down into water and oxygen. Oxygen is a gas and therefore wants to escape the liquid. However, the dish soap that you added to all your solutions is able to trap the gas bubbles, which results in the formation of a stable foam. As long as there is enzyme and hydrogen peroxide present in the solution, the reaction continues and foam is produced. Once one of both compounds is depleted, the product formation stops. If you do not add dish soap to the reaction, you will see bubbles generated but no stable foam formation.
If there is no hydrogen peroxide present, the catalase cannot function, which is why in cup one you shouldn't have seen any bubble or foam production. Only when hydrogen peroxide is available, the catalase reaction can take place as you probably observed in the other cups. In fact, the catalase reaction is dependent on the substrate concentration. If you have an excess of enzyme but not enough substrate, the reaction will be limited by the substrate availability. Once you add more hydrogen peroxide to the solution, the reaction rate will increase as more substrate molecules can collide with the enzyme, forming more product. The result is an increasing amount of foam produced in your cup as you increase the amount of H2O2 in your reaction. You should have seen more foam being produced once you added another tablespoon of hydrogen peroxide to cup one, which should have resulted in a similar amount of foam as in cup two. However, at some point you will reach a substrate concentration at which the enzyme gets saturated and becomes the limiting factor. In this case you have to add more enzyme to speed up the reaction again.
Many other factors affect the activity of enzymes as well. Most enzymes only function under optimal environmental conditions. If the pH or temperature deviates from these conditions too much, the enzyme reaction slows down significantly or does not work at all. You might have noticed that when doing the extra steps in the procedure.
Cleanup
Pour all the solutions into the sink and clean all the spoons with warm water and dish soap. Wipe your work area with a wet paper towel and wash your hands with water and soap.
More to explore
Biology for Kids; Enzymes, from Ducksters
Enzymes: The Little Molecules That Bake Bread, from Scientific American
Catalase, from PDB-101
Enzyme-Catalyzed Reactions—What Affects Their Rates?, from Science Buddies
The Liver: Helping Enzymes Help You!, from Scientific American
Science Activity for All Ages!, from Science Buddies
This activity brought to you in partnership with Science Buddies