Key concepts
Electricity
Batteries
Electrochemical reaction
Electric conductor
Introduction
Can you imagine how your life would change if batteries did not exist? If it were not for this handy way to store electrical energy, we would not be able to have all of our portable electronic devices, such as phones, tablets and laptop computers. So many other items—from remote-control cars to flashlights to hearing aids—would also need to be plugged into a wall outlet in order to function.
In 1800 Alessandro Volta invented the first battery, and scientists have been hard at work ever since improving previous designs. With all this work put into batteries and all the frustration you might have had coping with dead ones, it might surprise you that you can easily make one out of household materials. Try this activity and it might just charge your imagination!
On supporting science journalism
If you're enjoying this article, consider supporting our award-winning journalism by subscribing. By purchasing a subscription you are helping to ensure the future of impactful stories about the discoveries and ideas shaping our world today.
Background
Batteries are containers that store chemical energy, which can be converted to electrical energy—or what we call electricity. They depend on an electrochemical reaction to do this. The reaction typically occurs between two pieces of metal, called electrodes, and a liquid or paste, called an electrolyte. For a battery to work well, the electrodes must be made up of two different types of materials. This ensures one will react differently than the other with the electrolyte. This difference is what generates electricity. Connect the two electrodes with a material that can transport electricity well (called a conductor) and the chemical reactions fire up; the battery is generating electricity! As you make connections, note that electricity likes to take the path of least resistance. If there are multiple ways to go from one electrode to the other, the electricity will take the path that lets it flow most easily.
Now that you know the essentials of a battery, let's examine some household materials. Aluminum foil is a good conductor—electricity flows easily through it. The human body conducts electricity as well, but not as well as aluminum foil. Electrodes are as common as copper pennies you might have stashed in your piggy bank. As for electrolytes, they are found all over the kitchen; lemon juice is just one example. A simple household battery might be easier to make than you imagined!
Materials
At least two pennies
Water
A few drops of dishwashing soap
Paper towels
Aluminum foil (at least nine by 60 centimeters)
Scissors
Ruler
At least one lemon (preferably with a thin skin)
Plate
Knife (and an adult's help when using it)
At least two plastic-coated paper clips
Preparation
Wash your pennies in soapy water, then rinse and dry them off with a paper towel. This will remove any dirt sticking to them.
Carefully cut three aluminum foil rectangles, each three centimeters by 20 centimeters.
Fold each strip in thirds lengthwise to get three sturdy one-centimeter-by-20-centimeter aluminum strips.
Note: In this activity you will make a very low-voltage battery. The amount of electricity generated by this homemade battery is safe, and you will even be able to test it by touching your finger to it and feeling the weak current. Higher voltages of electricity, however, can be very dangerous and even deadly; you should not experiment with commercial batteries or wall outlets.
Procedure
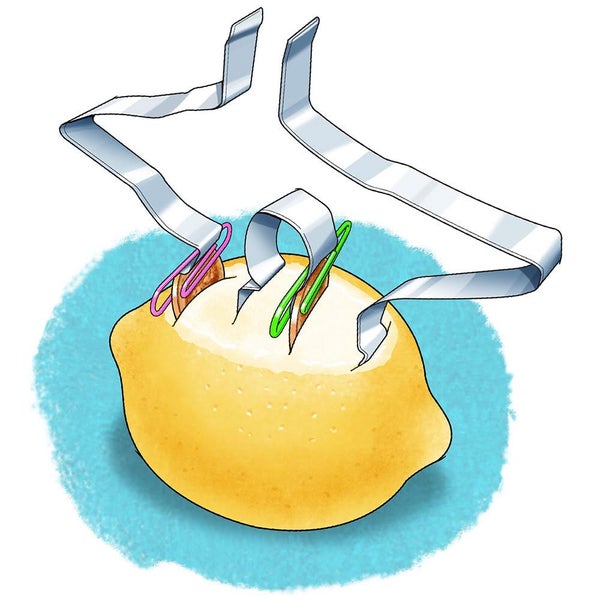
Place the lemon on its side on a plate and have an adult carefully use the knife to make a small cut near the middle of the lemon (away from either end). Make the cut about two centimeters long and one centimeter deep.
Make a second, similar cut about one centimeter away and parallel to the first cut.
Push a penny in the first cut until only half of it is showing above the lemon skin. Part of the penny should be in contact with the lemon juice because that is what serves as the electrolyte. This copper penny in contact with the lemon juice serves as your first electrode. Note: If your lemon has a very thick skin, you might need an adult to carefully cut away some lemon peel. Why do you think is it important for part of the penny to be in contact with the lemon juice?
Slide one of the aluminum strips in the second cut until you are sure part of the aluminum is in contact with the lemon juice. Can you guess which part of a battery the aluminum strip that sits inside the lemon is? Do you think it is important for the aluminum to be in contact with the lemon juice?
You have just made a battery! It has two electrodes made of different metals and an electrolyte separating them. Do you think this battery is generating electricity or is there still something missing?
Your battery can generate electricity but will only do so when the electrodes are connected with something that conducts electricity. To make a connection attach the second aluminum strip to the part of the penny sticking out of the lemon with a plastic-coated paper clip. Make sure the aluminum touches the penny so electricity can pass between the copper and aluminum. You used an aluminum strip to create a connection; would you expect a plastic strip to work as well? Do you know why you do not need to create a connection to the second electrode for this particular battery?
As soon as the two aluminum strips touch one another, electricity will be produced in the battery and flow through the strips, from one electrode to the other. Because you cannot see the electricity flowing, you can try to feel it. Keep the two strips about one centimeter apart and touch your fingertip to them. Can you feel a tingling, created by a small amount of electricity running from one aluminum strip to the other through your body?
For more electrical juice (and slightly stronger tingling sensation), you can build a second battery, identical to the first. You can choose a different spot on the lemon you just used or use a second lemon to build a second battery. Note that you only need one aluminum strip to build a second battery. To connect the second one to the original find the aluminum strip of the first battery that serves as electrode. (It has its end inserted in the lemon.) Use a plastic-coated paper clip to attach the other end of this aluminum strip to the penny of the second battery. This connects the aluminum electrode of the first battery to the copper electrode of the second battery.
Test this set of connected batteries in a similar way as you tested the single battery, bringing the ends of the two aluminum foil strips sticking out of your battery set (those that have a free end) in contact with your fingertip. Can you feel electricity running? If you could feel it well the first time, is this any different? (Note: If you cannot feel the tingling sensation, check if each electrode—pennies and the aluminum strips stuck in the lemon—are inserted deep enough so they are in contact with lemon juice; make sure there is firm contact between the penny and its attached aluminum strip; and that the aluminum strips are not touching one another. If all is correct, maybe you need slightly more electricity to feel tingling. You can test another person to see if he or she can feel the electricity or you can opt to add one more lemon battery to your set.)
Extra: Now that you can detect whether electricity is generated or not, try some different configurations. What happens if you let the aluminum strips touch? What happens if you replace an aluminum strip with a plastic piece, an unfolded metal paper clip or a toothpick?
Extra: Scientists call the way you connected your batteries in this activity "connecting batteries in series." Do you think the way you connect two batteries makes a difference in the amount of electricity you felt? Try it out by connecting the two copper electrodes to one another and attaching the two aluminum electrodes in the same way. (Note: You will need an extra strip of aluminum to do this.) Scientists call this "connecting batteries in parallel." Test both ways of connecting batteries and compare. Do you feel a difference?
Extra: Try different types of metals as electrodes for your batteries. Do you think a battery with two pennies as electrodes would generate electricity? What about a battery with a penny and a nickel? Note that some combinations might generate electricity but the amount generated might be below your ability to feel it. Connecting two or more of these batteries might help you identify good combinations.
Extra: You used a lemon to provide the electrolyte for your battery. Do you think other vegetables or fruits would work as well?Would a potato, apple or onion battery work? Try a few from around the kitchen (with permission, of course). Does one particular fruit or vegetable outperform the others? With what you learned about how batteries generate electricity, why do you think that one type of produce made a stronger battery?
Extra: If you have an LED (light-emitting diode) available, investigate how many lemon batteries are needed to light it.
[break]
Observations and results
Did you feel the tingling in your fingertip?
The battery you just made has a copper and an aluminum electrode separated by electrolyte lemon juice. It will generate electricity as soon as the electricity has a path to flow from one electrode to the other. You created this path using strips of aluminum, a material that conducts electricity well.
By connecting your battery to your fingertip, you allowed the small amount of electricity it generates to run through your body. This amount of electricity can create a tingling feeling in a fingertip. Experiences will differ from person to person. Some people might only feel the bigger signal generated by connecting several batteries in a particular way. Letting the aluminum strips touch provides a very easy way for the electricity to run from one electrode to the other, so almost no electricity will travel through your body and the tingling sensation disappears. Plastic and wood do not conduct electricity well; none will be felt when using these materials as connections. Metals, on the other hand, conduct electricity well. Different combinations of metals as electrodes will influence the amount of electricity generated. Using identical metals as electrodes will not generate electricity, however.
In this activity you made a very low-voltage homemade battery. But using commercial batteries can be dangerous—and never experiment with wall outlets!
More to explore
Batteries, from ExplainThatStuff!
How Do Batteries Work?, from LiveScience
A Battery That Makes Cents, from Science Buddies
Potato Batteries: How to Turn Produce into Veggie Power!, from Science Buddies
This activity brought to you in partnership with Science Buddies