Sickle cell disease, sleep disorders and lung cancer: these are just a few of the medical issues that were under investigation in at least 383 clinical trials that have had research grants terminated by the National Institutes of Health since February.
That’s about 1 in 30 of all the clinical trials—tests of medical interventions in human volunteers—funded by the federal agency, which has a $48-billion research budget, according to a JAMA Internal Medicine study published Monday.
The full list of 383 shelved clinical trials cited in the JAMA paper, obtained by Scientific American, reveals a wide range. The cuts follow a Trump administration drive to slash costs and cull funding for studies “misaligned” with its priorities; some 74,000 study participants have been affected by the trial cuts, according to the study.
On supporting science journalism
If you're enjoying this article, consider supporting our award-winning journalism by subscribing. By purchasing a subscription you are helping to ensure the future of impactful stories about the discoveries and ideas shaping our world today.
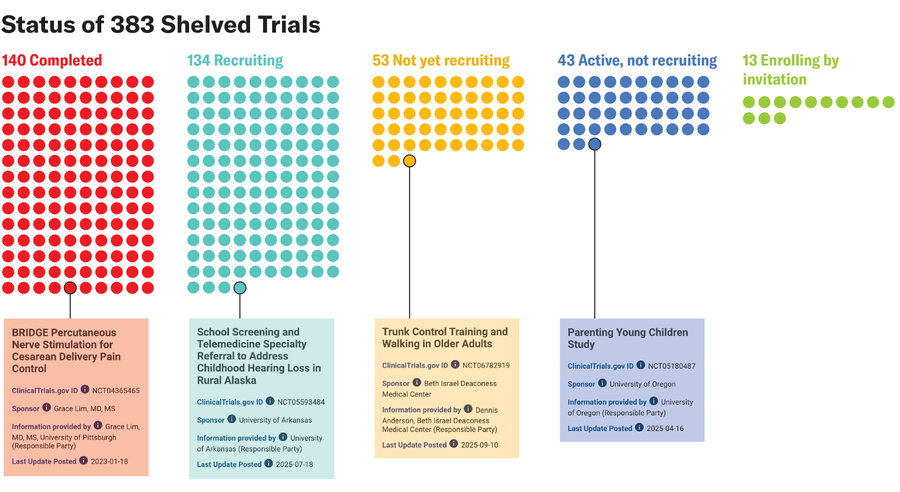
Amanda Montañez; Source: “Clinical Trials Affected by Research Grant Terminations at the National Institutes of Health,” by Vishal R. Patel et al., in JAMA Internal Medicine; Published online November 17, 2025 (data); clinicaltrials.gov (trial information)
“Trials involving cancer, cardiovascular disease, infectious diseases, mental health conditions, and other major sources of illness were among those affected,” says study co-author Vishal Patel, a physician at Brigham and Women’s Hospital.
“The range of conditions represented in the affected group shows that the impact was not limited to a narrow corner of research. It included areas where many patients face substantial health burdens,” Patel adds.
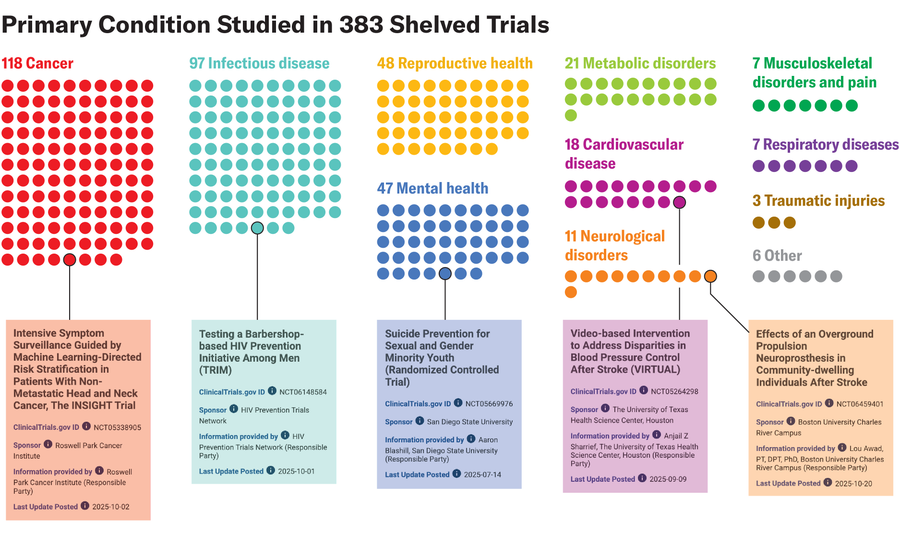
Amanda Montañez; Source: “Clinical Trials Affected by Research Grant Terminations at the National Institutes of Health,” by Vishal R. Patel et al., in JAMA Internal Medicine; Published online November 17, 2025 (data); clinicaltrials.gov (trial information)
In a statement, spokesman Andrew Nixon said the Department of Health and Human Services “strongly” rejects the study’s findings.
“NIH currently funds and oversees more than 42,500 active, recruiting or planned clinical trials across every major disease area,” he said.
“Selective focus on a handful of appropriately paused studies does not change that fact, and it should not be used to cast doubt on the overwhelming majority of trials that meet or exceed the gold standard of clinical research,” he said. “We are committed to ensuring that taxpayer dollars support programs rooted in evidence-based practices and gold standard science—not driven by ideological agendas.”
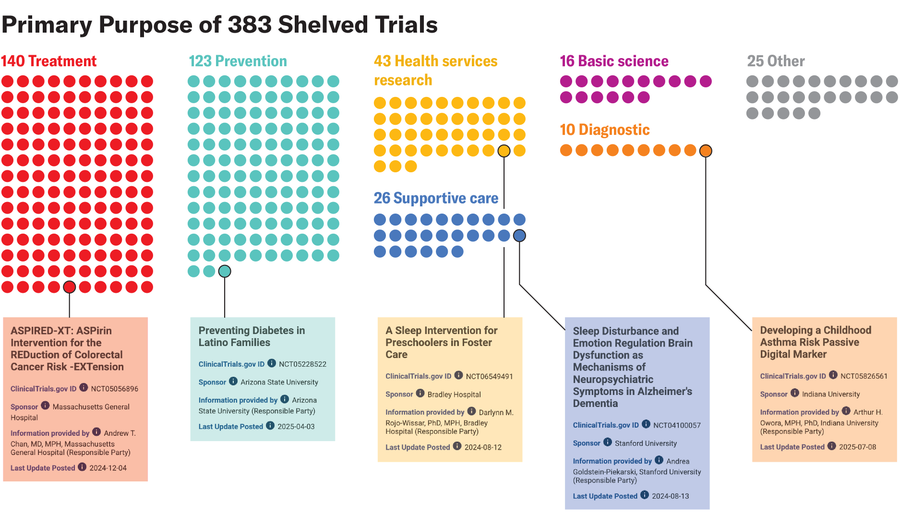
Amanda Montañez; Source: “Clinical Trials Affected by Research Grant Terminations at the National Institutes of Health,” by Vishal R. Patel et al., in JAMA Internal Medicine; Published online November 17, 2025 (data); clinicaltrials.gov (trial information)
People volunteer to join clinical trials for many reasons, whether out of a desire to help science or to access a new, promising drug. Everyone who participates in a trial exposes themselves to risk, but canceling the trials can also cause harm, says epidemiologist Bruce Psaty of the University of Washington.
“The intrusion of politics into the NIH with such a profound revision of its funding decisions is not among the traditional ethical reasons for stopping clinical trials early,” says Psaty, after reviewing some of the list.
“This intervention not only broke the original promise to participants of contributing to medical knowledge but it also subjected the participants, investigators, and their institutions to potential unanticipated harms,” he says.
Editor’s Note: This story is in development and may be updated.

