An investigational Alzheimer’s treatment from Eli Lilly failed to slow the progression of the memory-destroying disease in a late-stage trial, marking another setback in a field that has long bedeviled the drug industry.
The injected therapy, called solanezumab, didn’t meaningfully beat a placebo in a study on more than 2,100 patients with mild to moderate forms of Alzheimer’s, Lilly said. This marks the third time Lilly’s treatment has missed the mark in a late-stage trial, and the company has abandoned any plans to submit it for Food and Drug Administration approval.
The results “were not what we had hoped for, and we are disappointed for the millions of people waiting for a potential disease-modifying treatment for Alzheimer’s disease,” Lilly CEO John Lechleiter said in a statement. Lilly’s share price fell more than 14 percent on the news.
On supporting science journalism
If you're enjoying this article, consider supporting our award-winning journalism by subscribing. By purchasing a subscription you are helping to ensure the future of impactful stories about the discoveries and ideas shaping our world today.
The company spent hundreds of millions on solanezumab, testing and retesting it on ever narrower populations of Alzheimer’s patients in hopes of seeing a benefit. The drug is now among the dozens of would-be Alzheimer’s treatments waylaid by clinical disappointment. Over the past decade, roughly 99 percent of experimental drugs for the disease have failed in clinical trials, while the number of Americans with Alzheimer’s has risen to more than 5 million.
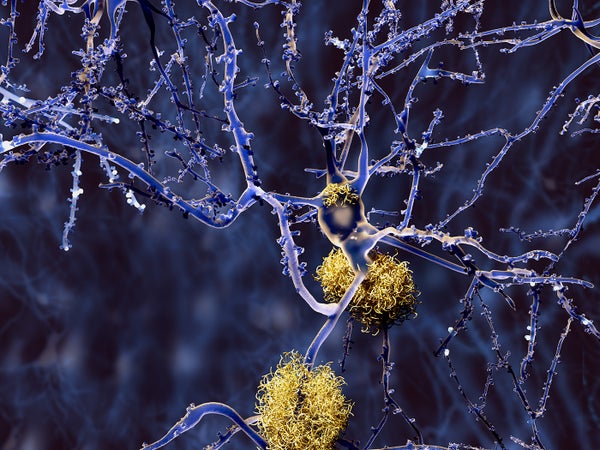
And Lilly’s failure could have a chilling effect on the entire field. Solanezumab is designed to sweep away plaques called beta-amyloids, which build up in the brains of patients with Alzheimer’s. Many scientists believe those plaques are key to the disease’s effect on neurons, but with each failed trial, that hypothesis gets harder to defend.
With Lilly’s drug sidelined, Alzheimer’s researchers will turn their attention to Merck, which is expected to present data in June on a drug that takes an upstream approach to amyloid. Called verubecestat, Merck’s treatment targets an enzyme called BACE, which regulates the body’s production of amyloid. By blocking BACE, scientists figure, they can cut off the flow of toxic plaques before they take effect.
After that, Biogen is slated to disclose late-stage data on an amyloid antibody that scientists and Wall Street analysts say could be the most promising treatment in its class. The drug showed tentative promise in a small study last year, creating big expectations for data from larger trials.
And Lilly isn’t giving up on Alzheimer’s. The company has a BACE inhibitor of its own in late-stage development in partnership with AstraZeneca, and it’s progressing a handful of early-stage therapies aimed at other Alzheimer’s targets.
Republished with permission from STAT. This article originally appeared on November 23, 2016