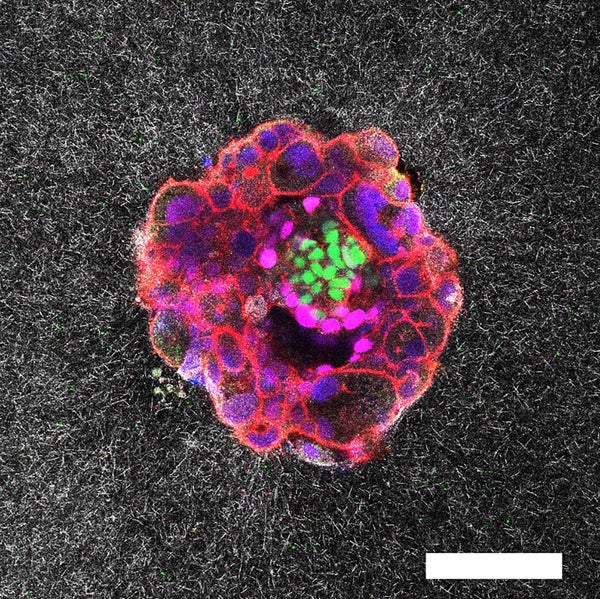
Researchers have captured the very first real-time, three-dimensional images and videos of a human embryo implanting into synthetic uterine tissue—revealing a key stage in reproduction. The resulting footage, which shows in vivid detail how embryos push and pull to anchor themselves in the uterus, could lead to improvements for in vitro fertilization techniques, the scientists say.
“This will allow us to develop treatments specifically targeting implantation, which is the biggest roadblock in human reproduction,” says Samuel Ojosnegros, a bioengineer at the Barcelona Institute of Science and Technology and a co-author of the new study in Science Advances.
A few days after an embryo is fertilized artificially, fertility doctors must implant it into the body so it can continue to grow. “What happens between the transfer and the first ultrasound weeks later is a black box,” says Ojosnegros, who is also co-founder of biotech company Serabiotics. Implantation failure is one of the main causes of infertility—up to 60 percent of miscarriages occur during this process.
On supporting science journalism
If you're enjoying this article, consider supporting our award-winning journalism by subscribing. By purchasing a subscription you are helping to ensure the future of impactful stories about the discoveries and ideas shaping our world today.
The first successful cultures of human embryos beyond the moment of implantation were demonstrated in laboratory petri dishes in 2016, but Ojosnegros and his colleagues wanted to see what this process would look like in 3D tissue that was more similar to that of the uterus.
To do this, the team designed a special system made of gel and collagen, a protein found in the uterine lining, and introduced embryos donated by people who had completed an assisted reproduction process. Their system prompted implantation, Ojosnegros says, because the network of collagen fibers signals the expected location and texture to the embryo at a molecular level.
The researchers recorded the action over time using advanced 3D microscopes. Tracking tiny movements in the gel’s fibers let them map exactly where and how strongly the embryos were pulling on the surrounding tissue. They then did the same with mouse embryos to compare their movement patterns.
The footage showed that human embryos generate a network of tiny pulling forces that ripple through the womb. These embryos burrow into the surrounding tissue by creating multiple small traction points that tug the lining in all directions. Mouse embryos, in contrast, spread out more across the surface and pull mainly along two or three strong lines.
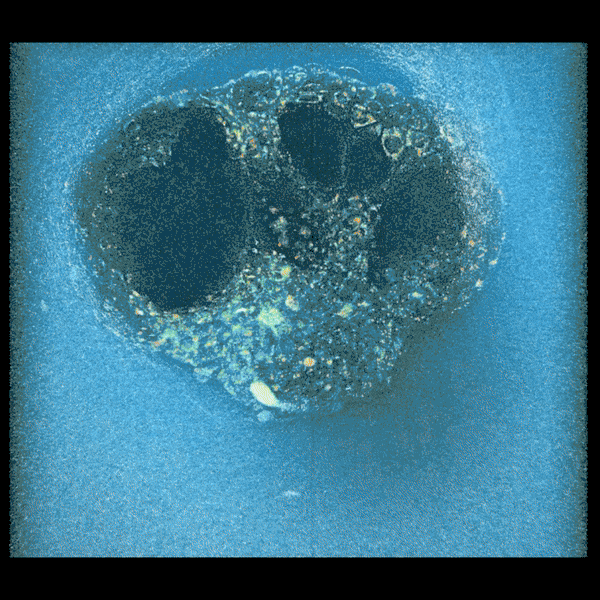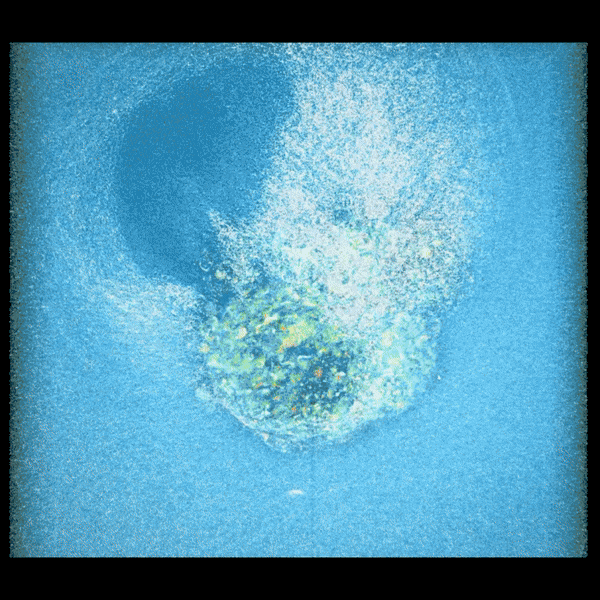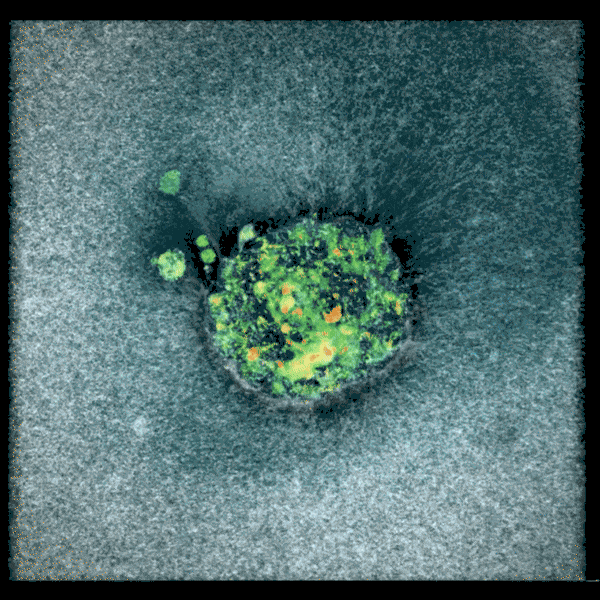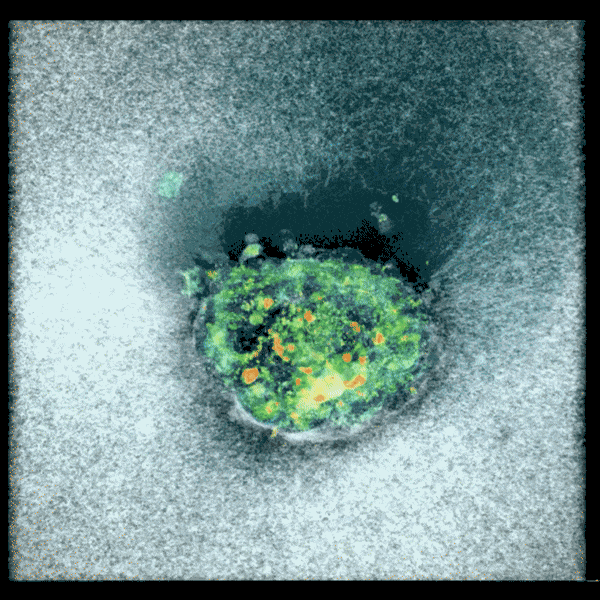
An embryo compacting and invading the uterine tissue.
Sarah Moreira Castro
When the researchers applied external tension to the matrix, tugging it with tiny forceps, they noticed the embryos reoriented toward those areas. The scientists suggest microcontractions in the uterus might direct the embryo to the optimal implantation surface. “We believe these microcontractions are what the embryo uses to guide itself toward the blood vessels and the nutrients it needs,” Ojosnegros explains, adding that more studies are needed to support this hypothesis.
In both mouse and human experiments, the strength and pattern of the pulling forces were linked to the embryos’ potential success, meaning embryos that pulled less were less likely to invade the tissue. Observing implantation in real time in a 3D model is a “quantum leap” compared with the two-dimensional observations that already exist, says developmental biologist Claudia Spits of the Free University of Brussels, who was also not involved in the new study. Keeping an embryo alive under these conditions is extremely difficult, she says. “What you see in a 10-second video is years of setting these [conditions] up so that the embryo can survive,” Spits adds.
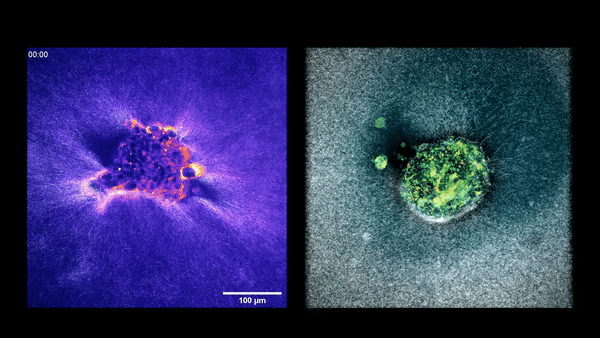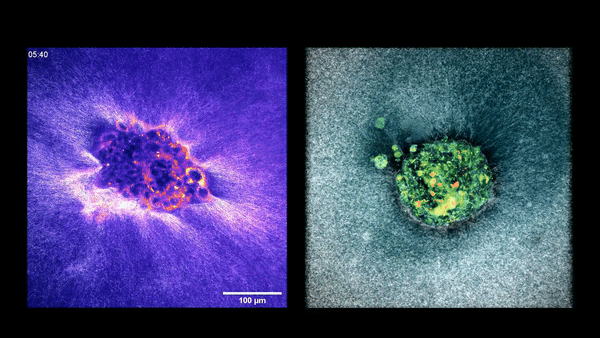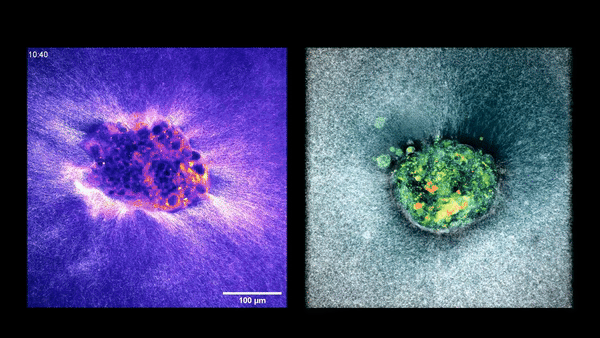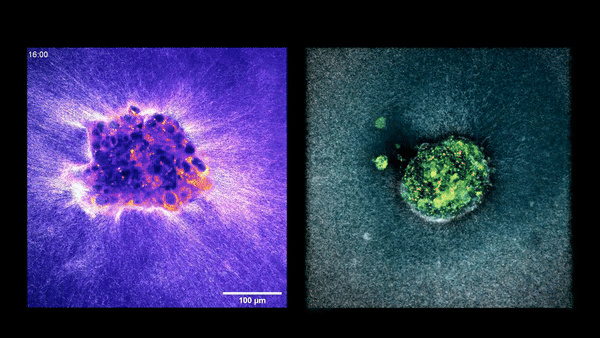
Two embryos implanting in the uterus.
Sarah Moreira Castro
“This study sets the stage to explore the dynamics of implantation in unprecedented detail,” says Magdalena Żernicka-Goetz, a developmental biologist at the California Institute of Technology, who was not involved in the research. The findings add to the growing library of human postimplantation observations published in the past nine years, she says, and “these studies are a thrilling step forward in understanding a stage of human development that has long been hidden from view.” Future research, Żernicka-Goetz notes, is still needed to compare how embryos behave across different “uteruslike” platforms to see whether developmental trajectories differ.
The matrix developed by Ojosnegros’s team is not intended for in vitro fertilization procedures, but it could be a valuable tool for pharmaceutical company and lab testing. “By beginning to understand how the embryo behaves,” Ojosnegros says, “we can start thinking about the future possibility of selecting healthy embryos or those more capable of implanting.” Spits remains skeptical of that assertion because replicating this technology in other labs could be a major challenge. But she says the results are a “major step forward” in tech that could have future applications once other labs are able to do their own 3D implantations.