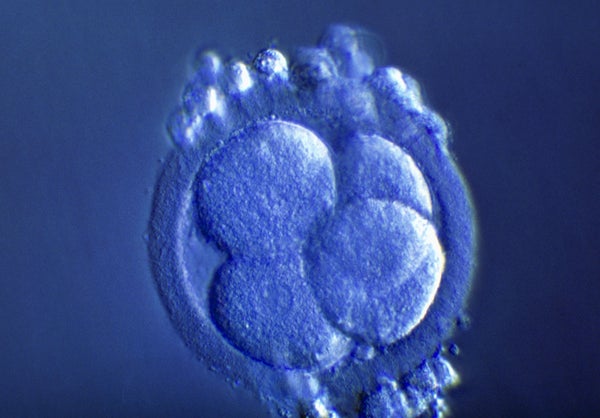
The international body representing stem-cell scientists has torn up a decades-old limit on the length of time that scientists should grow human embryos in the lab, giving more leeway to researchers who are studying human development and disease.
Previously, the International Society for Stem Cell Research (ISSCR) recommended that scientists culture human embryos for no more than two weeks after fertilization. But on 26 May, the society said it was relaxing this famous limit, known as the ‘14-day rule’. Rather than replace or extend the limit, the ISSCR now suggests that studies proposing to grow human embryos beyond the two-week mark be considered on a case-by-case basis, and be subjected to several phases of review to determine at what point the experiments must be stopped.
The ISSCR made this change and others to its guidelines for biomedical research in response to rapid advances in the field, including the ability to create embryo-like structures from human stem cells. In addition to relaxing the ‘14-day rule’, for instance, the group advises against editing genes in human embryos until the safety of genome editing is better established.
On supporting science journalism
If you're enjoying this article, consider supporting our award-winning journalism by subscribing. By purchasing a subscription you are helping to ensure the future of impactful stories about the discoveries and ideas shaping our world today.
“It’s been a major revision,” says Robin Lovell-Badge, a stem-cell biologist at the Francis Crick Institute in London and chair of the ISSCR steering committee that wrote the new guidelines.
Last revised in 2016, the document offers a rubric for what science the biomedical community agrees is worthy, and which projects are off-limits.
In the United States, where biomedical research involving stem cells or human embryos has been controversial for decades, and federal support has waxed and waned, the guidelines carry unusual weight, says Josephine Johnston, a bioethicist at the Hastings Center in Garrison, New York. Although US agencies have some policies covering such work, review committees at institutions or private funders often turn to the ISSCR’s document as the only regularly updated set of guidelines representing the views of the scientific community. “That means that when they make a change like this, it is actually fairly significant,” says Johnston.
The 14-day rule
First proposed in 1979, the 14-day rule bars research on embryos after they reach a key point of complexity. At least a dozen countries, including the United Kingdom, Canada and South Korea, have adopted the concept as law. Others, including the United States, have accepted it as a standard that guides researchers, reviewers and regulators.
With the new ISSCR recommmendations, Lovell-Badge envisions that the longer a researcher wants to culture an embryo for, the tougher the review process by a country’s regulatory authorities would be. “We’re not simply giving green lights for people to do this research,” he says. Furthermore, the guidelines say that public comment should be part of the review.
Before 2016, researchers weren’t able to keep human embryos alive in a dish for 14 days, so the rule didn’t bar any projects. But that year, two independent research teams announced that they had been able to grow human embryos in a dish for up to 13 days—they then terminated the experiments in accordance with the 14-day standard.
Such advances have led some ethicists and researchers to argue that the decades-old rule is antiquated and ripe for revision. Allowing embryos to grow past 14 days, researchers say, could produce a better understanding of human development, and enable scientists to learn why some pregnancies fail, for instance. The revised ISSCR guidelines are a prompt to begin conversations about when it would be valuable to grow embryos beyond 14 days, says Alta Charo, a bioethicist at the University of Wisconsin Law School in Madison, who was part of the ISSCR steering committee. “We didn’t debate it before—now it’s time to debate.”
Aryeh Warmflash, a stem-cell biologist at Rice University in Houston, Texas, believes weighing research benefits against ethical questions on a case-by-case basis, experiment by experiment, is an effective step—although he says he would eventually like to see more guidance on how to evaluate those trade-offs. But “it was a good choice not to frame this around advocating another ‘X-day’ rule”, he wrote in an e-mail to Nature.
Stem-cell model
In the past decade, scientists have made increasingly sophisticated models of embryos from human stem cells, demonstrating one way to study human development while avoiding the controversial use of embryos from fertility clinics. Such embryo-like structures are too rudimentary to grow into a person, scientists say. But relaxing the 14-day limit would allow researchers to compare them fully with real embryos, and test them as feasible stand-ins for research, says Lovell-Badge. Although the embryo-like structures are not technically bound by the 14-day rule, some scientists have said that they observe the limit when growing the model systems, because they are uncertain about the community consensus.
The relaxation of the 14-day rule “is really significant, but it’s done with a soft touch”, says Johnston.
Not everyone agrees that the shift is justified. Kirstin Matthews, a legal and policy scholar at Rice University’s Baker Institute for Public Policy in Houston, Texas, says there is unexplored science to be done with embryos that are two weeks or younger, and that given the public scrutiny of studies of human embryos, the ISSCR should have engaged the public while considering changes to the guidelines. “It doesn’t feel like we’ve exhausted our knowledge in this space,” she says.
Lovell-Badge acknowledges that the review and redrafting steps did not include public-engagement exercises, in part because of the cost and time involved. Also, an international public-comment period would probably receive varied responses from different jurisdictions, he says. “You’d have to make it a huge exercise, and we can’t do that.”
Shifts in genetic science
Some of the other key changes to the ISSCR’s ethics guide reflect advances in genetics.
For example, the guidelines now describe terms under which mitochondrial-replacement therapy could be used in medical research. Some metabolic diseases are caused by genetic mutations in the mitochondria, the power generators in cells, which children receive from their mothers. In cases where a mother’s mitochondria carry these mutations, doctors can now swap the nucleus from the mother’s egg cell into a donor cell with healthy mitochondria, whose nucleus has been removed, before in vitro fertilization (IVF). A baby born as a result of this technique would have mitochondrial genes from the donor, but their nuclear DNA would come from the mother and from the father whose sperm is used in IVF.
In 2016, US physician John Zhang announced that he had attempted such a procedure, and delivered in Mexico what news reports called a ‘three-parent baby’. At the time, some researchers worried that the country was chosen for its lax regulations. Since then, researchers in the United Kingdom have won approval to begin clinical trials of the method. In the United States, a clause in the annual budget legislation prohibits the Food and Drug Administration from considering such a technique, but Johnston says that might change soon. “I would be very surprised if it stays.”
The ISSCR guide also weighs in on whether it’s okay to edit the genes of human embryos or egg or sperm cells intended for implantation, and concludes that this science is still too risky. In 2018, scientists were alarmed by an announcement from Chinese biophysicist He Jiankui that he had used CRISPR–Cas9 technology to edit genes in human embryos that he then implanted in a woman’s uterus, resulting in the birth of twin girls. Since then, other expert panels have debated how to regulate gene editing that introduces heritable changes. They have pointed out that the procedure, still fairly nascent, can cause unintended changes to genes and has other technical flaws.
The ISSCR allows that the concept might be valuable in the future, for scientifically defensible reasons, once the science has advanced, and after extensive review. “As a matter of absolute principle, we do not say that heritable editing is absolutely wrong in every possible circumstance,” says Charo.
This article is reproduced with permission and was first published on May 26 2021.