From Tom Wolfe’s account of the LSD-loving Merry Pranksters and their bus ride across America in The Electric Kool-Aid Acid Test to Roger Sterling's acid-inspired bout of introspection on television's Mad Men, popular culture is rife with depictions of the mind-altering effects of lysergic acid diethylamide.
The synthetic drug, devised by Swiss chemist Albert Hofmann in 1938, pings some of the same receptors as the neurotransmitter serotonin, producing powerful changes in consciousness. But although the florid hallucinations and other effects of LSD are well known, their neurobiological bases have been less clear, partly because of restrictions placed on the drug after its recreational heyday in the 1960s.
New studies—prompted by a renewed interest in potential applications of psychedelic drugs for understanding the brain or even treating some psychiatric diseases—suggest that far-reaching changes in brain connectivity contribute to the altered states of consciousness and other effects of an acid trip. The latest work paints a picture of LSD and some other hallucinogens as drugs that can decrease modularity and connectivity within brain networks while enhancing the brain's overall connectivity, explains Frederick Barrett, a cognitive neuroscientist at Johns Hopkins University who has studied hallucinogenic drug effects but was not involved in the research released this week.
On supporting science journalism
If you're enjoying this article, consider supporting our award-winning journalism by subscribing. By purchasing a subscription you are helping to ensure the future of impactful stories about the discoveries and ideas shaping our world today.
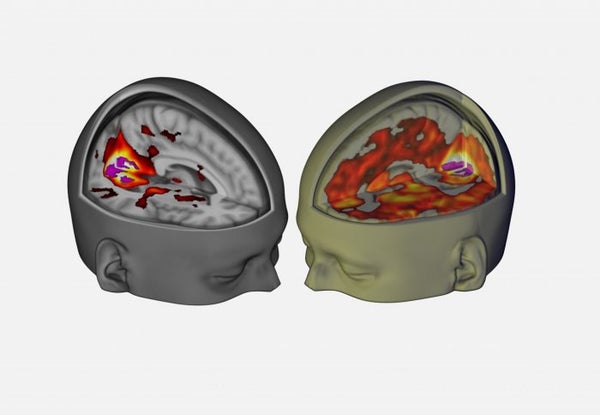
An Imperial College London–led team reporting this week in Current Biology and Proceedings of the National Academy of Sciences used brain imaging, electrical activity measurements and subjective reports to track brain and experiential responses to LSD in 20 volunteers. Results from the studies suggest that LSD simultaneously creates hyper-connections across the brain, allowing the functions of seemingly unrelated regions of the organ to ooze into one another. At the same time, the drug apparently chips away at organization within networks—including a system the brain defers to at rest called the default mode network, which normally governs functions such as self-reflection, autobiographical memory and mental “time travel.”
Members of the team reporting in Current Biology suspect that the default mode network disintegration, coupled with dampened electrical activity in consciousness-related alpha brain waves, contributes to a temporary loss of a sense of self in some psychedelic drug users, who often describe feeling at one with others and the world around them—an effect scientists call “ego dissolution.”
The default mode network “seems to be the best candidate that we have for the biological underpinnings of the sense of self,” explains Robin Carhart-Harris, a researcher with Imperial College’s Center for Neuropsychopharmacology, who was involved in both new studies. Similar default mode network changes were identified in a study of 30 volunteers who took an intravenous dose of another psychedelic drug: psilocybin, the psychoactive ingredient in hallucinogenic, or “magic,” mushrooms. Carhart-Harris and colleagues published that study in PNAS in 2012.
Changes in connectivity have also come forth as prime candidates for LSD’s ability to produce visual hallucinations. In their recent PNAS paper members of the Imperial College team reported that the presence of LSD was linked to a boost in blood flow and functional connectivity between the brain's primary visual cortex and seemingly unrelated brain networks that mediate other sensory inputs as well as higher-level processes including emotion. These connections were enhanced in users who reported intense, dreamlike visions.
To uncover these and other neural consequences of LSD, Carhart-Harris and his colleagues recruited 20 volunteers by word of mouth, selecting prior psychedelic drug users to minimize the risk of frightening effects. "When we're putting people in a brain scanner—particularly an MRI scanner, which is quite noisy, quite confined, quite claustrophobic—it's important to know that people are going to be able to tolerate that environment," Carhart-Harris notes.
Participants came to the lab twice, two or more weeks apart. During one session they received a 75-microgram intravenous dose of LSD, roughly equivalent to one "hit" for recreational acid-droppers. For the other session, researchers administered a saline solution placebo intravenously. At roughly 100 minutes after each treatment, the team began testing brain activity in participants at rest with their eyes closed, first using a form of functional magnetic resonance imaging (fMRI) called arterial spin labeling to follow blood flow changes in the brain. Next, the resting participants underwent two scans with another fMRI method known as blood oxygen level–dependent, or BOLD, to track connections and apparent information flow between brain regions. Finally, nearly four hours into the acid (or placebo) trip, the team did two rounds of magnetoencephalography scanning to assess the brain's electrical activity.
During the scans research subjects used a push-button system to report the LSD effects they experienced. In questionnaires completed immediately after the test and again weeks later users recounted the extent of their hallucinations, level of altered consciousness, the longer-term significance of their experiences, and other details. The researchers discarded data for five participants who moved too much for accurate scans to be obtained. But data for the remaining 15 let them look at connectivity and other brain effects, aligning them with the specific experiences that the LSD users described.
Although the current studies were conducted on seasoned psychedelic trippers, Carhart-Harris suspects the same general neurobiological patterns would turn up in individuals who were new to the drugs. That remains to be tested experimentally—if studies involving first-time LSD users are someday approved.
For their part, Carhart-Harris and his team are keen to study other types of psychedelic drugs, including dimethyltryptamine (DMT), sometimes found in a traditional South American brew called ayahuasca. They have also done a pilot study on psilocybin as a therapeutic option in individuals with treatment-resistant depression, and are interested in possible roles for psychedelics in combating mental illnesses or addiction.