Key concepts
Physics
Forces
Molecules
Surface tension
Introduction
Have you ever wondered how water droplets form on a window on a rainy day, why some bugs can walk on water or why you use soap to wash your hands? All of these things are related to the phenomenon called surface tension. In this activity you will learn how you can use surface tension to make a fish cut from a piece of paper seem to swim around on its own!
Background
Whether riding in a car on a rainy day or spilling water on the kitchen counter, you have probably seen water droplets form. What holds these water droplets together—sometimes in nearly spherical shapes? Why don’t they spread out evenly or fall apart into even smaller drops? They are held together by something called surface tension. Water molecules are slightly attracted to one another; at the surface of a water droplet, they are more attracted to the other molecules around them than to the air above. This forms a thin “skin” of strongly attracted water molecules that are difficult to break through.
On supporting science journalism
If you're enjoying this article, consider supporting our award-winning journalism by subscribing. By purchasing a subscription you are helping to ensure the future of impactful stories about the discoveries and ideas shaping our world today.
Sometimes high surface tension can be beneficial—such as for insects that walk on water. Other times we want to lower water’s surface tension—for instance, when we are doing laundry or washing dishes. This is because there are lots of tiny holes in between pieces of dirt or clothing fabric, and high surface tension can make it hard for water to seep into those holes. Adding soap or detergent decreases water’s surface tension, making it easier to penetrate all those little nooks and crannies. Do you think you can make use of this change in surface tension to make an object skim across the surface of the water? Try this project to find out!
Materials
Shallow tray
Tap water
Piece of paper
Scissors
Medicine dropper
Soap
Workspace that can tolerate soapy water spills
Preparation
Cut out a small “fish” shape from your piece of paper. Make sure the fish has a V-shaped notch in its tail.
Fill your shallow tray partially with water and bring it to your workspace.
Procedure
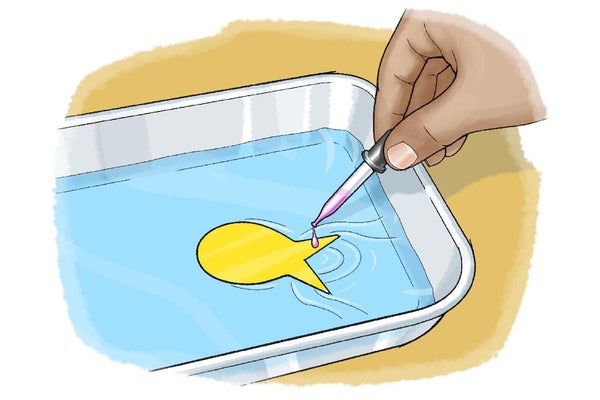
Carefully place the paper fish on the surface of the water at one end of the tray with its tail facing outward and its head facing into the tray. Make sure the fish floats and you do not push it underwater.
Use the medicine dropper to carefully place a single drop of soap on the water in the V-shaped notch of the fish's tail. What happens?
Empty, rinse and dry the tray; cut out a new paper fish and try again. Do you get the same result?
Try reusing one of the paper fish with fresh water, reusing the tray of water with a new paper fish or both. Can you get it to work a second time? Why or why not?
Extra: Try cutting out and testing some different shapes. Do the shapes have to be “streamlined” like a fish? What if the shape is symmetric like a circle or blocky like a square?
Extra: Try the activity with different liquids you have in your kitchen, such as vegetable oil or juice. Do any liquids have the same effect as soap? Do any of them not work? Why do you think this happens?
Observations and results
When you place the paper fish on the water’s surface and it floats, the surface tension of the water pulls at it from all sides. Because it is completely surrounded by water, surface tension pulls evenly in every direction, and it does not move. When you add a drop of soap to one side of the fish, the surface tension on that side suddenly becomes lower so the fish is not pulled as hard in that direction, and it moves in the other direction. This is a great example of Newton's first law of motion! The fish is initially at rest and the forces on it are balanced so it remains at rest. When you add the soap, the forces on it become unbalanced so it accelerates.
You will probably find it is difficult to either reuse a paper fish or a tray of water. Once the fish and/or water are “contaminated” with some soap, the soap spreads and the surface tension is lowered everywhere, rather than on just one side of the fish, so you can no longer make it move. You can probably find, however, the effect does work with many other shapes—even squares! You might also observe that other liquids, such as oils (which have a lower surface tension than water) work just as well as soap, but liquids such as juices (which are mostly water) do not.
Cleanup
Clean up any spilled liquids.
More to explore
Surface Tension, from Hyperphysics
Build a Raft Powered by Surface Tension, from Science Buddies
Measure Surface Tension with a Penny, from Scientific American
Science Activities for All Ages!, from Science Buddies