For the first time, researchers have obtained images of individual carbon dioxide molecules trapped in a series of molecular “cages”—and they borrowed an imaging technique from biologists to do it. Metal-organic frameworks (MOFs) are exceptionally porous polymers designed to capture particular gas molecules, letting scientists separate or purify various vapors. Even small amounts can slurp up a lot of gas: a single gram can have a gas-grabbing surface area nearly the size of two football fields. MOFs have been proposed for holding hydrogen in automobile tanks or fuel cells (without the need for extra cooling) and for grabbing and storing planet-warming carbon dioxide emissions, among many other uses.
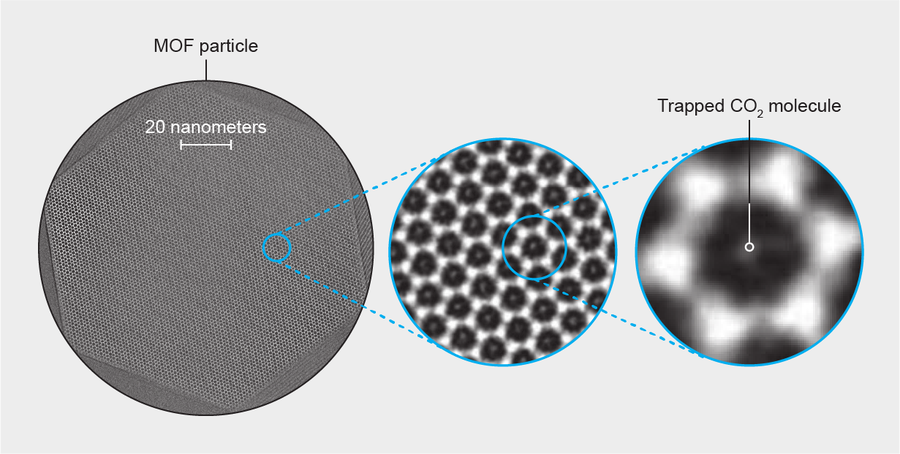
Credit: Amanda Montañez; Source: “Cryo-em Structures of Atomic Surfaces and Host-Guest Chemistry in Metal-Organic Frameworks,” by Yuzhang Li et al., in Matter, Vol. 1, No. 2; August 7, 2019
When Yuzhang Li, a materials scientist at Stanford University, and his colleagues examined a sample of a CO2-trapping MOF with a transmission electron microscope, they found the instrument's powerful electron beam “just melted” the honeycomblike framework, Li says. So the researchers tried an approach that biologists often turn to when imaging delicate proteins, viruses and cell organelles: they used liquid nitrogen to freeze and stabilize the material at a nippy −170 degrees Celsius and also dialed back the strength of their electron beam. This method let them take long-exposure pictures—not only of a slice through the material itself, called ZIF-8 (top and middle) but of the CO2 molecules trapped within it (bottom). The team reported its results in the August issue of Matter.
On supporting science journalism
If you're enjoying this article, consider supporting our award-winning journalism by subscribing. By purchasing a subscription you are helping to ensure the future of impactful stories about the discoveries and ideas shaping our world today.
This flash-freezing process will allow detailed studies of how MOFs trap gas, says Jeffrey Long, a materials chemist at the University of California, Berkeley, who was not involved in the study. For example, Li says, future work might generate 3-D images to investigate how quickly and efficiently the materials pull in gases.

