Lithium-ion batteries are at the heart of nearly every electric vehicle, laptop and smartphone, and they are essential to storing renewable energy in the face of the climate emergency. But all of the world’s current mining operations cannot extract enough lithium and other key minerals to meet skyrocketing demand for these batteries. Establishing new mines is an expensive, years-long effort. And mining also creates a host of environmental headaches—such as depleting local water resources and polluting the nearby region with runoff debris—that have led to protests against new mines.
All of this means the ability to recycle existing batteries is crucial for sustainably shifting the global energy system. But recycling lithium-ion batteries has only recently made commercial inroads. Battery manufacturers have hesitated over concerns that recycled products may be lower in quality than those built from newly mined minerals, potentially leading to shorter battery life or damage to the battery’s innards. Consequences could be serious, particularly in an application such as an electric vehicle.
But new research published in Joule has hit upon what experts describe as a more elegant recycling method that refurbishes the cathode—the carefully crafted crystal that is the lithium-ion battery’s most expensive component and key to supplying the proper voltage. The researchers found that batteries they made with their new cathode-recycling technique perform just as well as those with a cathode made from scratch. In fact, batteries with the recycled cathode both last longer and charge faster. The team’s approach and successful demonstration are “very unique and very impressive,” says Kang Xu, an electrochemist at the U.S. Army Research Laboratory, who was not involved in the study.
On supporting science journalism
If you're enjoying this article, consider supporting our award-winning journalism by subscribing. By purchasing a subscription you are helping to ensure the future of impactful stories about the discoveries and ideas shaping our world today.
A Joke No More
Yan Wang, a materials science professor at Worcester Polytechnic Institute and co-author of the new study, started researching battery recycling 11 years ago. At the time, he says, “some people joked with me, ‘There’s not enough batteries for you to recycle.’” That joke is not aging well. The Department of Energy estimates the battery market may grow 10-fold over the next decade. To ease the market’s growing pains, “recycling of lithium-ion batteries—getting that material back into the supply chain—is critical,” says Dave Howell, director of the DOE’s Vehicle Technologies Office. The DOE funded the new research as part of its massive effort to spur large-scale battery recycling innovations in the U.S.
When a lithium-ion battery is providing power, a cluster of lithium ions moves from one crystalline “cage” (the anode) to another (the cathode). The most common methods currently used to recycle these batteries involve dismantling and shredding the whole battery, then either melting it all down or dissolving it in acid. The result is a black mass—with a texture can that can vary from powder to goo—from which chemical elements or simple compounds can be salvaged. Those recovered products can then go through the same commercial manufacturing process that newly mined elements do to make cathodes.
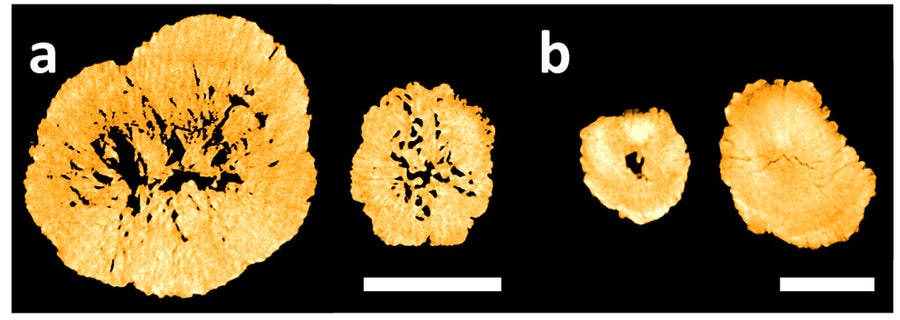
Cross sections of the recycled cathode particles (A) and particles made from new materials (B), taken with an X-ray microscope. The scale bar is 10 micrometers in (A) and 5 micrometers in (B). Credit: “Recycled cathode materials enabled superior performance for lithium-ion batteries,” by Xiaotu Ma, et al., in Joule, Vol. 5, No. 11; Nov. 17, 2021 DOI:https://doi.org/10.1016/j.joule.2021.09.005
Wang and his colleagues use a very similar process—but instead of completely breaking the battery down to its constituent chemical elements, their technique keeps some of the old cathode’s crucial composition intact. After they shred the battery, they physically remove the less expensive bits (such as the electronic circuits and steel battery casing) and recycle them separately. What is left is mainly the cathode material; they dissolve this in acid and then remove impurities. Next, they carefully add just a touch of fresh elements that compose the cathode, such as nickel and cobalt, to ensure the ratio of ingredients is just right—another distinction from common recycling methods. After a few more steps, the result is an effectively refreshed cathode powder, composed of tiny crystalline particles that can be stuck onto a metal strip and placed in a “new” battery.
Because a cathode is crafted from a precise mix of precious minerals to achieve the battery’s specific voltage, slight changes to its structure or composition can compromise its performance. Thus, much of the cathode powder’s value is “in how you’ve engineered the particles [of powder] in the first place,” says Emma Kendrick, a professor of energy materials at England’s University of Birmingham, who was not involved in the new study. That value is lost if the entire battery is simply melted down or dissolved in one fell swoop, as in current recycling methods.
More Pores, Faster Charge
Wang and his colleagues compared the particles in their recycled cathode powder with those in commercially manufactured cathode powder (largely made from newly mined minerals). They found that the recycled powder particles were more porous, with particularly large voids in the center of each one. These characteristics provide room for the cathode crystal to swell slightly as lithium ions squeeze into it, and this wiggle room keeps the crystal from cracking as easily as cathodes built from scratch. Such cracking is a major cause of battery degradation over time.
More pores also mean more exposed surface area, where the chemical reactions that are necessary to charge the battery can happen—and this is why Wang’s recycled batteries charge faster than their commercially manufactured counterparts. A future ambition could be to design all cathodes to have this superior structure rather than just those made from recycled stuff, Wang says.
The latest findings demonstrate that “the cathode they can make is as good as—or even better than—the commercial material that we’ve been importing,” says Linda Gaines, a transportation analyst at Argonne National Laboratory and chief scientist at ReCell Center, an organization that studies and promotes battery recycling. (Gaines was not involved in the new study.) Such imports largely come from China, which leads the world in battery recycling. But this situation means materials must be shuffled across the globe to be recycled, increasing the carbon footprint of recycled batteries and diminishing their allure as a more sustainable path. The approach developed by Wang’s team cuts out a significant chunk of international trade and transportation requirements, carving a potential path for other countries to bolster domestic battery recycling. The process is currently being scaled up by Ascend Elements, formerly Battery Resourcers, a recycling company Wang co-founded.

