There’s a plate of cookies on my desk. I put it there myself, so I can only blame myself for avoiding work with the prospect of a sweet snack. I don’t even want to know exactly how many calories there are in a single little cookie. Yet this is exactly what you should know if you want to lose weight.
The calorie is a unit of energy and is actually out-of-date. The official measure should be given in joules. But especially when it comes to energy that we add to our body by ingesting food, we still like to use the calorie. The same applies to mathematical formulas such as the basal metabolic rate, the amount of energy the body uses at rest for basic functions, including breathing and staying warm:
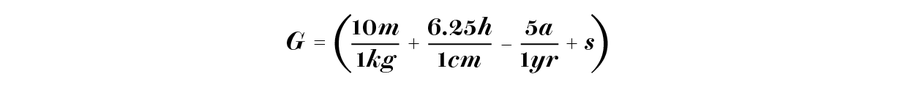
On supporting science journalism
If you're enjoying this article, consider supporting our award-winning journalism by subscribing. By purchasing a subscription you are helping to ensure the future of impactful stories about the discoveries and ideas shaping our world today.
If you insert the weight m (in kilograms), the height h (in centimeters) and the age a (in years) and choose the factor s according to gender (+5 for men and –161 for women), you get the Mifflin–St. Jeor formula that shows the basal metabolic rate in kilocalories per day. This is the amount of energy that our body needs to keep up its basic functions.
The value from the formula is not exact. In reality, the basal metabolic rate depends on many other factors than just weight, height and age. Everyone has a different metabolism, and hardly anyone lies around motionless or is solely concerned with maintaining basic bodily functions. Any kind of activity increases energy expenditure. Only in certain cases of bedridden sick people does the basal metabolic rate approximate the actual energy requirement.
Nevertheless, it is good to know just how much energy you can ingest if you want to lose weight. To do so, the basic rule is that you have to use more energy than you consume. And if you want to avoid the yo-yo effect—endless cycles of losing weight loss and then gaining it all back—you should make sure that the change in the energy balance is reasonably sustainable.
The many different diets in various magazines are generally of little help, even if they try to impress you with the math, as in the case of the “minus 500” formula. If you believe the puffery, then it is considered to be an ingenious formula. It is thought to be an equation developed by nutrition experts, a “mathematically proved” strategy to lose weight without all the problems that one usually has with diets.
Minus 500 turns up frequently in weight-loss programs. The formula is simple: multiply body weight in kilograms by 30 and subtract 500, and the result of the calculation is the amount of kilocalories that you can consume per day. And as much as I like to rant about it, this formula actually works. That is because body weight times 30 is a simple estimate for the basal metabolic rate. It is, of course, much less precise than the Mifflin–St. Jeor formula, but it is not drawn completely out of thin air. And if you subtract 500 kilocalories from your basal metabolic rate and limit your daily energy intake to this value, you will definitely consume less energy than you use.
From a purely mathematical point of view, there is nothing wrong with the minus 500 formula. But the question of whether it is always appropriate to reduce your energy intake in this simple way should be checked with a medical professional beforehand. In the end, however, I have to grumble a little. The math is merely giving weight to a simple fact. In the end, the “ingenious formula” developed by the “experts” only comes down to what everyone actually knows: if you want to lose weight, you have to eat less and exercise more—which is why my cookie and I are going to take a walk.
This article originally appeared on Spektrum.de and was reproduced with permission.

