Summer sunlight flickers through warm waters off the coast of Spain. The sea looks calm and peaceful. Near the surface, invisible to the naked eye, a swarm of microscopic plankton, some orange-pink and others dark green, swims in lazy circles, capturing the sun’s rays and using the solar energy to make nutrients through photosynthesis. Suddenly, a tentacled creature called Mesodinium—at 22 microns, a giant next to some of the three-micron sun-gathering plankton—comes zigzagging through the waters, drawn by sugars and amino acids leaking from the smaller organisms. Its tentacles shoot out and engulf the hapless green prey, or nanoflagellates, which are completely consumed and digested.
The predator is pickier, though equally brutal, with its pink-colored prey, called cryptophytes. While it digests and destroys most pieces, the attacker takes in the organelles responsible for photosynthesis whole. Within minutes the pale Mesodinium starts to turn darker red as it fills its body with the stolen parts—chloroplasts and nucleosomes—which remain intact and functional. Mesodinium is not able to take in and use carbon dioxide as true photosynthesizers are, so it relies on its victims’ chloroplasts to accomplish that task. The creature’s dual strategy of hunting for food like an animal and photosynthesizing like a plant is known as mixotrophy.
Mesodinium does not, however, get to hold onto its loot for long. Nearby lurks yet another mixotroph, slightly larger and with different hunting skills: the dinoflagellate Dinophysis. It starts circling the other creature and then shoots out harpoonlike threads that immobilize the Mesodinium. The captor then administers the coup de grâce. It lances its target with an appendage called a peduncle, something that looks and acts like a straw, and sucks out the innards, including the stolen chloroplasts. These now third-hand photosynthetic factories are assimilated into the new host and begin churning away within the Dinophysis, giving it energy for life. Remnants of the original thief, the butchered Mesodinium, drift away.
On supporting science journalism
If you're enjoying this article, consider supporting our award-winning journalism by subscribing. By purchasing a subscription you are helping to ensure the future of impactful stories about the discoveries and ideas shaping our world today.
These single-celled killers are only two examples of countless billions of mixotrophic plankton that swim in our seas. For a long time, most marine scientists dismissed mixotrophs as minor curiosities, compared with the two main groups of single-celled plankton that are supposed to dominate the base of the ocean’s food web. One of these groups, plantlike phytoplankton, uses light energy and inorganic nutrients such as nitrates to proliferate. The other group, animallike zooplankton, eats these phytoplankton. In this way, nutrients are passed up the food web to bigger animals. Next to these two purists, mixotrophs were deemed to be inefficient oddballs, jacks-of-all-trades but masters of none. (There are some rare examples of mixotrophs on land, such as the carnivorous Venus flytrap plants.)
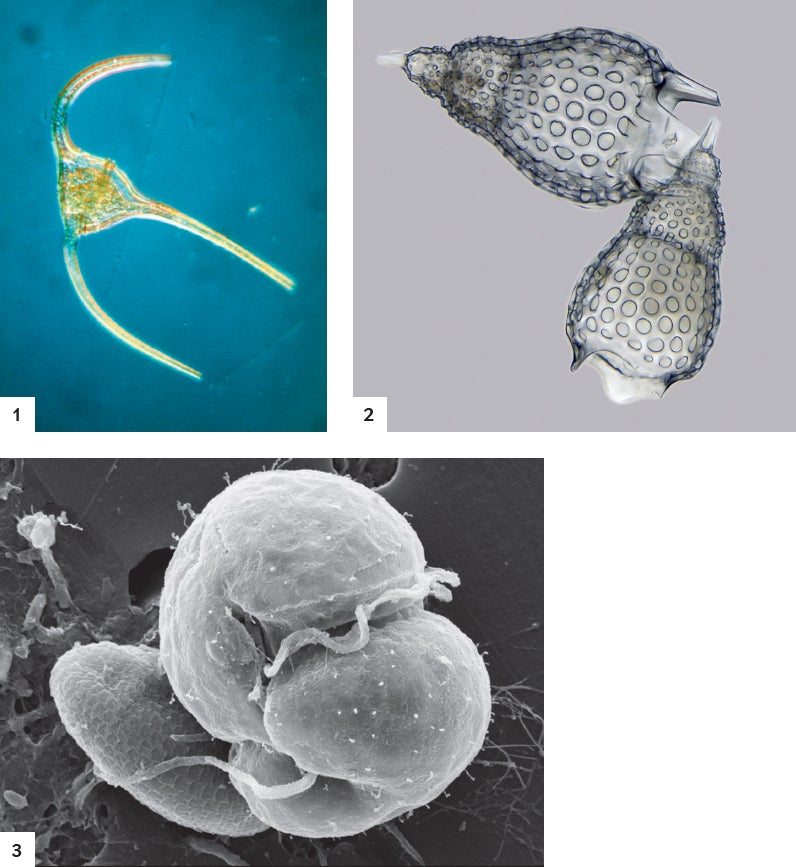
Tiny Monsters: Mixotrophs come in several varieties. Some, such as Tripos longipes (1), can photosynthesize on their own and eat prey. But members of the order Nassellaria (2) steal photosynthetic organs from plankton victims. Species of Karlodinium (3) act as T. longipes does. Credit: Eric Grave Science Source (1); Frank Fox mikro-foto.de (2); Vincent Lovko Mote Marine Laboratory (3)
This traditional view of the ocean food web is wrong. Through experiments, observations and models of plankton populations, my colleagues and I have recently uncovered evidence that most single-celled plankton are neither purely plantlike nor pure plant eaters. The great majority are, in fact, mixotrophs. This means the bottom of the food web—and thus everything above it—is not controlled the way we thought it was. If most plankton are really mixotrophs, their numbers are not sharply limited by photosynthesis but can increase through eating. And solar energy, when available, can give an extra boost to eating-driven growth. These abilities have a ripple effect on many things, from the atmosphere to fish populations. Greater mixotroph activity, for instance, affects how quickly the oceans remove climate-warming carbon dioxide from seawater and air and lock it away in sea-bottom sediments. Mixotroph groups may be less susceptible to the waxing and waning of sunlight as seasons change. Such versatility and resilience have advantages. Populations of beneficial mixotrophs can provide sustenance for more fish larvae and increase the human food supply. But there can be negative consequences, too. Some mixotroph species cause harmful algal blooms that close shellfish hatcheries and lead to widespread fish kills.
If this new view of marine biology holds water—as recent findings by ourselves and other scientists have shown—it means the idea of an ocean ecology based on “plantlike” and “animallike” plankton is no longer tenable. There is a different, strange and powerful player amid the waves.
Attack of the Triffids
Mixotrophs are like something out of science fiction. The harpoons and peduncles of Dinophysis seem, on a smaller scale, like features of the planet-conquering plants in the famous 1951 sci-fi novel The Day of the Triffids, by John Wyndham. Triffids could use their roots to extract nutrients from the soil and to walk around. But they also had venomous stingers that they used like whips, blinding or killing humans and then feasting on the decomposing bodies.
The first time I heard of plankton mixotrophs was a little more than a decade ago, during my Ph.D. research on microzooplankton, which are smaller forms of animallike plankton. (The smaller plantlike organisms, likewise, are called microphytoplankton.) Textbooks described mixotrophs as a weird oceanic rarity. Yet with their dual ability to photosynthesize and hunt, combined in a single cell, mixotrophs appeared to be nature’s perfect beasts. Because evolution tends to favor efficiency, it surprised me that mixotrophy was not more common. Searching for more information about it, I found a whole suite of exciting publications by Diane Stoecker, a plankton ecologist at the University of Maryland’s Horn Point Laboratory, whose field and laboratory work suggested that mixotrophy existed among ocean plankton. I got in touch with Stoecker, and our discussions convinced me that more mixotrophs were out there. But how many, and what were they doing?
My own expertise is in building mathematical models of food webs to understand the behavior of different organisms in them. In essence, these are simulations that run on a computer. In searching through ocean ecosystem models, I could not find one that simulated the details of mixotrophs’ double lives. Nor could I find funding for a project to build one; scientific review committees did not think mixotrophs were very important. So I spent my days working as a biodiversity officer for the local government in Bridgend, Wales, and in the evenings I worked on the model with my marine biologist husband, Kevin Flynn. By the spring of 2009 we had a working simulation, one that could represent different populations of mixotrophs, some hunting more and some doing more photosynthesis. It was published that year in the Journal of Plankton Research.
Our goal was to show that a model including dual-natured plankton could simulate marine ecology more realistically than other models that segregated ocean populations into predators and plants. We changed mixotroph characteristics in the model until we had simulations that captured real-life observations of nutrient flow within food webs, as well as interactions among other plankton types such as bacteria and tiny crustaceans called copepods. These food web dynamics, which we published in 2010 in the Journal of Marine Systems, were very different from the segregated plankton models.
We needed to go beyond computer simulations, however. We had to gather evidence supporting our hypothesis that mixotrophs were critical drivers of the ebb and flow of nutrients through all parts of the ocean and all creatures in it. This time there was money. Impressed by the success of our models, a foundation called the Leverhulme Trust supported a series of meetings in Europe and the U.S., where for the first time, researchers who worked with mixotrophs in the field and the lab could share what they knew.
A World of Hybrids
At the first meeting in 2011, our group (we dubbed ourselves “Team Mixotroph”) made a list of all the plankton species known to both hunt and photosynthesize. Scientists had, over the course of decades, identified mixotroph species in water samples across all marine systems, from coasts to midocean areas and from the poles to the equator. They carried out experiments in labs onboard the research ships. And in some instances, they brought back the plankton to their institutes and carried out additional experiments using different nutrients, prey or light intensities to find out how the mixotrophs behaved under different environmental conditions. Until our group started combining these observations, most researchers thought they were looking at small and unusual occurrences rather than a common life-form throughout the world’s oceans.
.png?w=900)
Credit: Rebecca Gelernter; Source: “Defining Planktonic Protist Functional Groups on Mechanisms for Energy and Nutrient Acquisition: Incorporation of Diverse Mixotrophic Strategies,” by Aditee Mitra et al., in Protist, Vol. 167, No. 2; April 2016
Bringing these data together drove us to the conclusion that there was a lot of mixotrophy happening in the sea and that it was ecologically essential. For example, Per Juel Hansen, a plankton ecophysiologist at the University of Copenhagen, and his colleagues demonstrated that without sufficient cryptophyte prey (those pink plankton mentioned earlier), the Mesodinium population would not be able to acquire stolen chloroplasts and would die out. Stoecker and her team, as well as Hae Jin Jeong and his colleagues at Seoul National University, showed that when mixotrophs were actively photosynthesizing, they ate other plankton at higher rates than those that were not performing photosynthesis: one mode enhanced the other. And when light and nutrients were abundant, these particular mixotrophs had a much higher growth rate than did plankton stuck with only a single process.
In 2012 we started looking beyond the mere presence of mixotrophs in marine ecosystems and began to identify different groups based on what they eat, how they eat and how they photosynthesize. It turns out there are four distinct types, and each occupies a different place along a spectrum of mixed behavior.
The first criterion for discriminating among different types of mixotrophs was to identify the source of their photosynthetic abilities. Did they have an inherent capacity for using light to make food, or did they have to attack and steal photosystems from their prey? We called the group with their own genetic capabilities to produce and maintain the body parts for photosynthesis constitutive mixotrophs. This group includes a lot of benign and ecologically important creatures that are critical links in ocean food chains. But it also contains plenty of troublemakers. We see their effects in destructive algal blooms when their populations get out of hand. For example, the mixotroph Karlodinium is renowned for causing massive fish kills across the world, from the Chesapeake Bay to Malaysian coastal waters. Prymnesium, another constitutive mixotroph that has caused similar fish kills off the coast of Texas and in the backwaters of the Norfolk Broads in England, releases a chemical that destroys the integrity of cell membranes belonging to competing plankton. As a result, these plankton swell up and explode. Then Prymnesium consumes the debris. Toxins produced by another species, Alexandrium, make their way into mollusks as those animals ingest plankton-laced water. Entire oyster, mussel and clam fisheries are then closed because humans who consume seafood contaminated with Alexandrium can get paralytic shellfish poisoning.
The second group does not have inherent photosynthetic capabilities and has to hijack them. These are called nonconstitutive mixotrophs and include Mesodinium and Dinophysis. This group is a big collective. Its members’ use of photosynthesis had been considered a supplemental survival mechanism employed only when there was not a lot of prey around. Now we realize that they harness solar energy much more often, and it is usually a crucial part of their way of life.
Credit: Jen Christiansen; Source: “Defining Planktonic Protist Functional Groups on Mechanisms for Energy and Nutrient Acquisition: Incorporation of Diverse Mixotrophic Strategies,” by Aditee Mitra et al., in Protist, Vol. 167, No. 2; April 2016
The nonconstitutive mixotrophs can be subdivided into generalist and specialist species. The plastidic ciliates Laboea and Strombidium, for instance, are generalists that steal chloroplasts from many different types of plankton. Generalists cannot maintain these looted parts for more than a few days and must constantly attack new prey for replacements. They tend to be helpful mixotrophs, contributing nutrients to food webs that support fisheries, and thus play a key role in global food security.
Specialists, on the other hand, depend solely on a particular type of prey and seem to be better equipped by evolution to integrate stolen photosystems into their own physiology; they can maintain their pilfered assets for weeks to months. Some, such as Dinophysis, can be harmful to humans. Shellfish exposed to Dinophysis can give people life-threatening food poisoning, and large blooms, such as some that have occurred in the Gulf of Mexico, have closed oyster farms.
Some specialists can be split off into yet another group with a remarkable kind of behavior. They do not just steal body parts, like Mesodinium does, but instead take in and enslave entire colonies of photosynthetic prey. The colonies live and proliferate within the host, feasting on nutrients and enjoying protection from outside predators. These planktonic greenhouses include microorganisms called foraminiferans and radiolarians and are found throughout the oceans. Over hundreds of millions of years, foraminiferans have played a vital role in the global biological carbon pump, taking in large amounts of the element, sequestering it when they sink to the seafloor and later releasing it in smaller amounts as they decompose. Analyzing ancient layered deposits of foraminiferans has helped us to assemble records of past climate changes and to link them to mass extinction events. Not all these floating greenhouses are harmless, however. One type, the green Noctiluca species, is capable of causing harmful algal blooms in polluted coastal waters.
Global Regulators
From plants that eat to animals that photosynthesize and from tiny two-micron organisms to relatively large one-millimeter plankton, mixotrophs span a wide range of ocean life. Why does this matter? Because it turns out that small organisms can have a variety of large impacts.
There is, for instance, an enormous nutrient-scarce area covering thousands of square kilometers in the middle of the central Atlantic Ocean. Scientists used to think that phytoplankton competed with marine bacteria for dissolved inorganic nutrients such as iron and phosphates in this area, which did not leave much to go around. But Mikhail Zubkov, a microbial biogeochemist at the National Oceanography Center in England, and his colleagues found a sizable population of constitutive mixotrophs—the ones that do their own photosynthesis—in this area when they sampled the waters during research cruises.
From these observations, Team Mixotroph developed two food web simulators. One was based on the traditional model of plant-bacteria competition, and the other added the mixotrophs. The team found that the mixotroph simulation was the one that best matched the nutrient amounts and cycles that Zubkov had observed. Instead of competing with plantlike phytoplankton, the bacteria grew using sugars and other nutrients that leaked out of the mixotrophs. Then the mixotrophs turned around and ate the bacteria, which gave them more phosphates and iron than they could pull from the seawater. And the model only fit observations if the mixotrophs were constitutive.
There was another key effect. With mixotrophs, and not traditional phytoplankton, the levels of carbon fixation—carbon dioxide taken out of seawater by the organisms—were significantly increased. The finding suggests that if these hybrids were not around, global amounts of carbon, which contribute to planetary warming, might be even higher.
Mixotrophs are especially important in coastal seas, where their effects on fisheries can be profound. In 2017, using a North Sea plankton model with mixotrophs of varying types, we found that when small mixotroph species eat marine bacteria, their populations grow larger, and as a result, they can outcompete other plankton that tend to form algal blooms. These scumlike blooms are not toxic, but they do block sunlight, putting a big crimp in a nutrient cycle that feeds tiny fish larvae and helps them to grow. Fewer blooms means more fish.
For fish health, it is also important that mixotrophs, as our observations have shown, turn out to be the dominant plankton form during summers. Pure plantlike plankton grow in the spring but then decline, so delicate fish larvae cannot depend on them. But mixotrophs are still around, and they are good, rich food that sustains the fish during this period.
A Dual Future
Mixotrophs are at the center of so much in marine science, be it climate change and fisheries projections, reconstructing ancient time lines of carbon cycling or predicting destructive algal blooms. The challenge now is to use both real-time observations and our models to establish what the different mixotroph groups are doing in different locations during different seasons. This is important because as our climate changes, we need to know which environmental conditions would lead to a bloom of the toxic Karlodinium, or the ecologically damaging, green Noctiluca, or the fisheries-sustaining plastidic ciliates. We have recently completed the first steps toward this goal, mapping the presence of different mixotroph groups across the global seas. We next need to measure population sizes during varying seasons because changing light and temperature drastically affect their growth and proliferation.
There are still marine scientists who point out these conclusions rest on our simulations as much as they rely on real-world observations, and that is a valid critique. It is why we need to get more scientists to examine mixotroph activity beyond the lab, out on the high seas.
In 2017 I applied to the European Commission for a grant to train scientists to do this work. In stark contrast to my fellowship application 10 years earlier, this grant was funded, with glowing comments from scientific reviewers. Our expanding Team Mixotroph will be able to bring the next generation of marine researchers up to speed. Together we hope to figure out the many ways in which the oceans’ perfect beasts may control our imperfect world.