This article was published in Scientific American’s former blog network and reflects the views of the author, not necessarily those of Scientific American
It seems like every couple of weeks I see a breaking headline announcing the discovery of the “holy grail” of battery technologies that will one day allow our smartphones to charge in a matter of minutes, enable inexpensive electric vehicles with a range of hundreds of miles, and allow cheap and reliable renewable energy storage.
Yet it seems like none of these “game-changing” batteries ever make it past the the headlines. So what’s the deal?
Venkat Viswanathan, an Assistant Professor in the Department of Mechanical Engineering at Carnegie Mellon University, asked the same question. In a recent paper, he and his colleagues developed methods to quantify a battery chemistry’s position in what they call the “Hype Cycle.”
On supporting science journalism
If you're enjoying this article, consider supporting our award-winning journalism by subscribing. By purchasing a subscription you are helping to ensure the future of impactful stories about the discoveries and ideas shaping our world today.
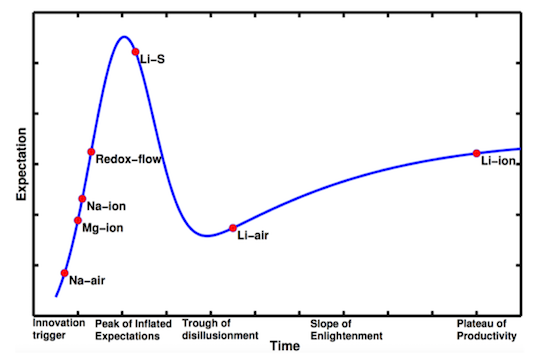
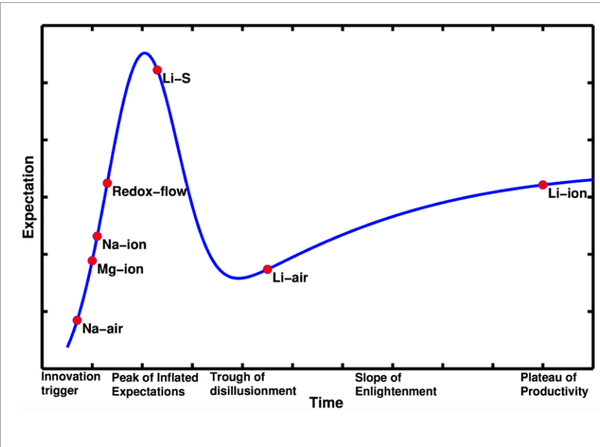
The researchers used the rate of new technical papers published about a particular battery chemistry to determine its position in what they call the Hype Cycle: ranging from the initial “innovation trigger” and “peak of inflated expectations” to the eventual “plateau of productivity,” where lithium-ion (Li-ion) batteries sit today.
The chart shows a few different classes of battery technologies. Magnesium-ion (Mg-ion), sodium-ion (Na-ion), and lithium-sulfur (Li-S) batteries all work kind of like Li-ion batteries do today, but they have the potential to beat Li-ion when it comes to energy density (i.e. how small/light the battery is) and cost. In particular, Li-S batteries have the potential to be drastically cheaper than conventional Li-ion batteries due to the low cost of sulfur. However, present Li-S batteries suffer from instabilities that cause a huge drop in efficiency and increased self discharge. Furthermore, Li-S battery electrodes can swell by as much as 80 percent as ions are shuttled around within the cell, so it’s difficult to design materials that can hold the battery together. Nevertheless, Li-S batteries are one of the most promising technologies for the future, having just traversed the “peak of inflated expectations” identified by Viswanathan and his students.
The only battery chemistry besides Li-ion to overcome the “trough of disillusionment” and approach the “slope of enlightenment” is lithium-air (Li-air). Li-air has enormous potential to beat out Li-ion because it uses a fundamentally different technique to store energy. The battery cell uses metallic lithium in its negative side and reacts with atmospheric oxygen on its positive side. Because one of the reactants in the battery is air, in theory you need half as much battery materials to store the same amount of energy, and the weight of the battery can be reduced by half. This is especially appealing for electric vehicles, which would benefit greatly from smaller battery packs. However, Li-air batteries still have a long way to go before they can achieve the cost and lifetime of conventional Li-ion batteries and reach reach the “plateau of productivity.”
Viswanathan’s study is useful because it helps to put the state of different battery technologies in perspective. While there are a lot of great innovations on the horizon, Li-ion is still the winning technology when it comes to portable electronics, electric vehicle, and grid applications. Perhaps in the next few years we’ll see Li-air catch up to Li-ion. That would make electric vehicles seriously competitive with their gasoline- and diesel-fueled counterparts. But Li-air still has a long “slope of enlightenment” ahead of it before it can really compete with Li-ion, let alone fossil fuels.
Read the full academic paper referenced in this post here: Oleg Sapunkov et al., 2015.