A human heart might contract some three billion times over a lifetime, and each one of those contractions is driven by a protein called myosin. One of three classes of so-called motor proteins in our cells, myosin converts chemical energy into movement.
Myosin usually moves to a steady beat, but like all biological mechanisms, its action is vulnerable to disease. And when the myosin in cardiac tissue malfunctions, the consequences can be devastating.
Collectively called cardiomyopathy, these disorders of heart muscle are thought to affect up to one in 500 people worldwide. It can leave them short of breath, fatigued after walking up a flight of stairs, with disconcerting, fluttering heartbeats and an almost constant feeling of being tired. Many sufferers end up making dramatic shifts in their life: giving up their favorite activities, changing to a less strenuous job, or even stopping work altogether.
For years there was little that could be done, but that is starting to change.
“We can think about how to address these diseases by designing therapies that improve the function of muscle,” says Fady Malik, head of research and development at the biopharmaceutical company Cytokinetics.
That’s a relatively new idea, he adds. “Traditionally, drug companies didn’t really think of muscle as a therapeutic target.”
Myosin modulators
That view changed over the last couple of decades as the basic science improved, including the discovery of both genetic causes of cardiomyopathy and molecules that can alter the activity of myosin and other motor proteins.
These developments have triggered the development of novel treatment options—specifically a new class of drugs called myosin modulators—for patients with heart muscle disorders, says Sharlene Day, a cardiac disease expertat the University of Pennsylvania School of Medicine. “That's been a very exciting development in our field, because before those drugs we could only address symptoms of disease.”
Researchers at Cytokinetics have played a key role in this new field since the late 1990s, when they started to look closely at the function of motor proteins. They originally hoped to treat some cancers by inhibiting a class of motor proteins called kinesins, which guide the transfer of genetic material during cell division. But then a surprising finding turned their thinking on its head.
“We wanted to inhibit kinesin, but we also found that we could activate it,” Malik says. That was a novel idea at the time. “There were not any small-molecule activators of proteins. There were certainly none for motor proteins.”
There was no clinically relevant reason to activate kinesin. But that didn’t apply to all motor proteins. “The aha moment was realizing that it might also work to activate myosin in the heart, and how that could be therapeutically relevant,” Malik explains.
It was a good time to switch attention to the heart. After decades of uncertainty, scientists around the world were starting to piece together the genetic causes of cardiomyopathy and how they could affect the functioning of myosin.
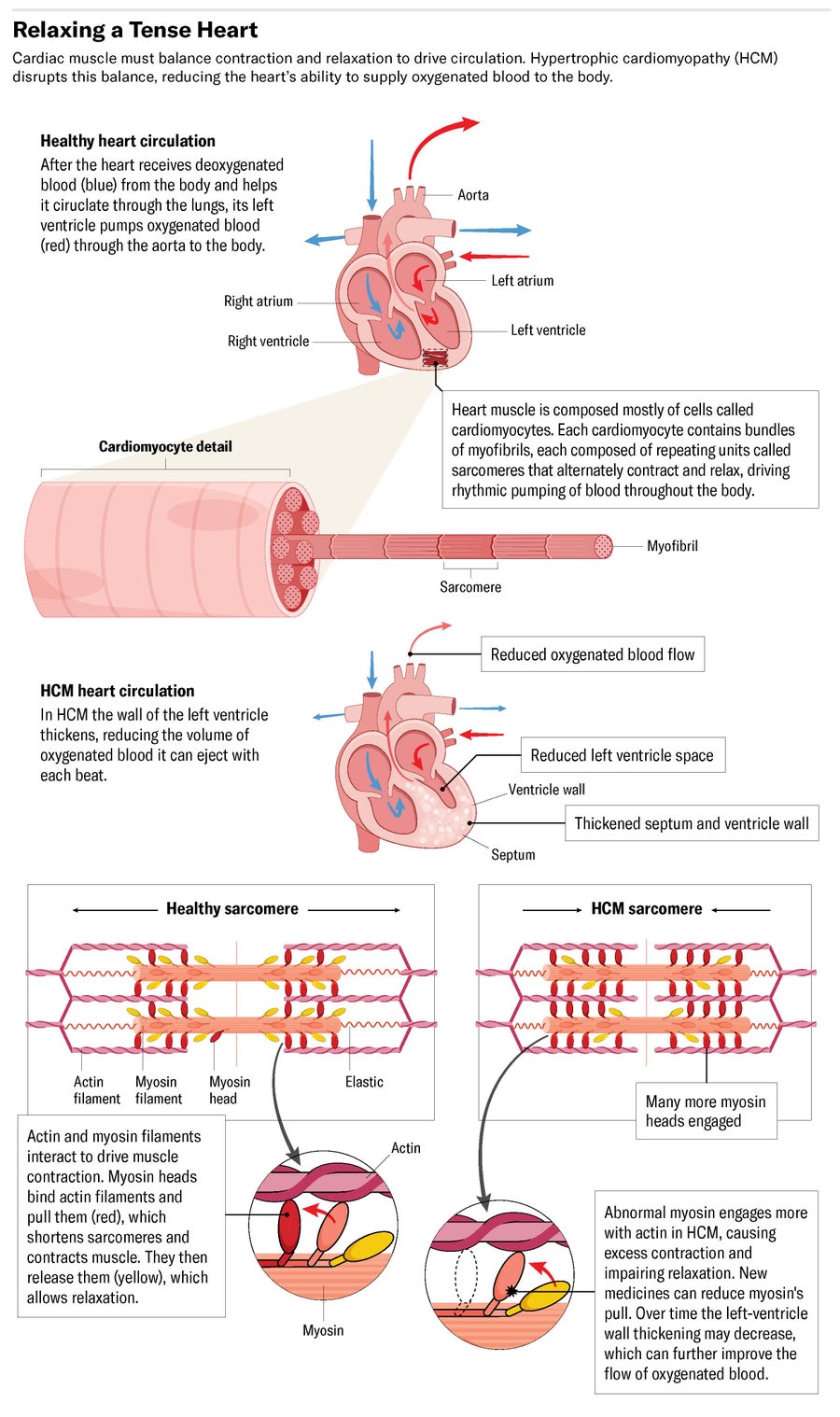
One important step was to link changes to myosin to the behavior of the sarcomere, the basic contractile unit of a muscle fiber. In a sarcomere, myosin binds filaments made of a separate protein called actin. By binding the actin filaments and flexing, myosin can make the sarcomere compress like a spring, then by letting go of the actin, it allows the spring to release. A single sarcomere generates a small force, but together the tens of thousands of sarcomeres that make up a muscle fiber work together to generate the powerful strokes needed for the heart to pump blood.
“That has to occur every second on the second for someone's lifetime. So, you can't have any interruption,” Day says. Genetic mutations in myosin can threaten this regularity, she explains, by making the protein fold differently or by disrupting binding sites. “That's where the problems arise.”
Genetic causes
Depending on what those genetic variants do to the protein, they can cause either a thickening of the muscle called hypertrophic cardiomyopathy, or thinning of the muscle and a subsequent decrease in force generated. “There's a lot of steps between the genetic variant and the cardiomyopathy,” she adds, which can make it difficult to understand the exact mechanisms involved.
That makes the sarcomere a key structure to investigate in the search for therapies for these diseases, and particularly the way that its enzymes process a fuel molecule called ATP that provides the energy needed to drive the actin-myosin interaction and therefore the heartbeat.
In theory, a drug that can activate this enzymatic use of ATP could address forms of cardiomyopathy in which the sarcomere contraction is too weak. But the sarcomere is a complicated structure. To find drugs that could affect its function, researchers needed to recreate that structure in a lab.
“It takes a lot of specialized knowledge to be able to purify the proteins of the sarcomere and put them back together,” says Malik. “And that's where we came in.”
Synthetic sarcomeres
By adapting laboratory techniques to collect sufficient protein and optimizing them, the Cytokinetics team built a simple synthetic sarcomere. That involved finding ways to blend separated myosin and actin back together in the right ratios and in sufficient quantities, but without the solutions becoming too viscous to work with.
After scaling up production of their synthetic sarcomeres, the scientists used them to screen hundreds of thousands of possible small compounds, looking for those that increased myosin activity based on how much it hydrolyzed ATP.
This screening process yielded basic molecules that could be used to investigate what impact a drug could have, says Jim Hartman, a muscle physiology researcher at the company. “And that gave us something we could put on a piece of muscle and have it squeeze more strongly and generate more force.”
“Over the last 15 years or so, it’s emerged that mutations in the sarcomere, some of them in myosin, actually make your heart squeeze too much,” Hartman says. Since myosin mutations could alter how much the heart squeezes, chemical compounds might as well. “Our concept was that if you work at the motor protein level, maybe we could treat different types of heart failure that have either too much or too little contractility.”
That’s possible, he adds, because, for a muscle that beats billions of times, cardiac tissue is unusually sensitive. “Your heart generally works in a pretty narrow range of performance. So small changes can make a big difference.”
Alisdair Macdonald
Explore how muscle function supports health, what goes wrong in disease, and how emerging therapies can help.



