The big goal for firms that are devising tests for Alzheimer’s is to find a simple, inexpensive one that is reliable enough to provide definitive diagnoses—something that could replace entirely the need for expensive PET scans and cerebrospinal fluid biopsies. Scientists at Toronto-based RetiSpec believe that retinal scans might eventually meet this bill.
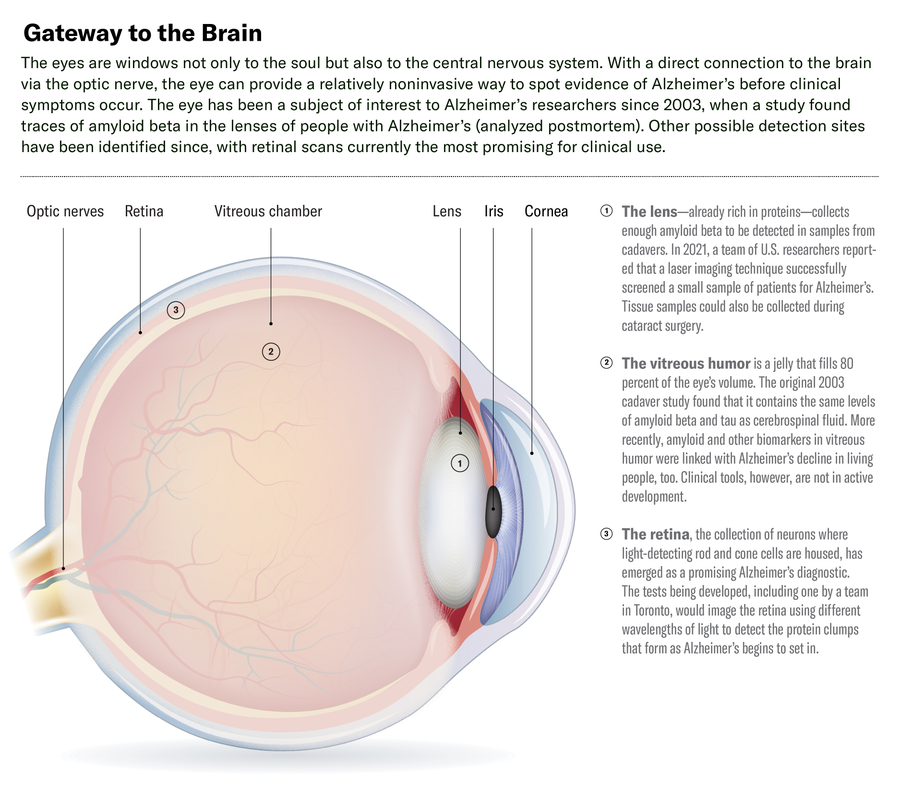
The eye is linked to the brain by way of the optic nerve, so changes in the brain—including the buildup of amyloid plaque from Alzheimer’s—are reflected in the retina. RetiSpec has developed a device that beams light on to the retina and measures the light reflected back. It then analyzes the output using AI algorithms trained on other retinal scans collected from thousands of Alzheimer’s patients who had tested positive on PET and cerebral spinal fluid tests.
The device is an attachment that can be screwed on to a device, called a fundus camera, that most eye and neurology specialists, and some primary care physicians, already have. Although the company has not yet set a price for the device, officials say that the cost per test will be very affordable to ensure broad accessibility. Whereas normal fundus cameras capture three wavelengths of color, red, green and blue, the attachment consists of a hyperspectral sensor that can capture a wider band of frequencies, most of which are not visible to the naked eye.
Hyperspectral sensors have detected Alzheimer’s disease in lab mice. CEO Eliav Shaked says that a multicenter, blinded validation study showed the company’s AI-based retinal test can predict amyloid-beta brain pathology in a diverse population of adults with preclinical, mild cognitive impairment, and probably Alzheimer’s disease, and that the study, led by the Global Alzheimer's Platform Foundation, is under review at a scientific journal.
RetiSpec hopes eventually to win FDA approval for use as a diagnostic tool, rather than just for purposes of screening. Retinal scans, he says, are more convenient than blood tests because they can provide a diagnosis immediately and don’t require the use of sensitive diagnostic equipment currently needed to analyze blood samples. The company is testing the device at 20 clinics and doctor’s offices. It is slated to begin feasibility testing with the FDA in early 2024. Shaked expects the technology will be available in early 2025.
Katie Peek
This article is part of The New Age of Alzheimer’s, a special report on the advances fueling hope for ending this devastating disease.