Alzheimer’s disease (AD) can be devastating for patients and their families and is a significant public health concern for society. Eisai’s commitment to Alzheimer’s drug discovery spans over four decades, culminating in the approval of multiple interventions, including a symptomatic treatment and an anti-amyloid monoclonal antibody. The development of novel disease-modifying therapies remains an active area of focus. Beyond drug discovery, Eisai is committed to advancing scientific understanding of AD, including characterization of AD biology to inform clinical practice.
Multiple smoldering pathologies
AD is driven by a constellation of pathological changes that together result in progressive clinical symptoms first appearing as mild impairment, followed by worsening dementia1. When Alois Alzheimer discovered AD in 1906, he identified the two core pathological changes of amyloid-beta (Aβ) plaques and neurofibrillary tau tangles (NFTs) in the brain. Following more than one hundred years of research, we know that other changes, including neurodegeneration, inflammation, and vascular brain injury, also contribute to the onset of clinical symptomology characteristic of AD1. This network of interconnected pathologies spans multiple brain regions, collectively fueling the pathophysiological underpinnings of smoldering AD.
In a broad range of other disease states, the term “smoldering” has been used to describe a constellation of pathophysiological processes that drive gradual disease progression. For example, in multiple sclerosis (MS), much of the groundwork for this concept originated from studies on anti-inflammatory therapies, which showed that a significant reduction in lesions (also called plaques) was not sufficient to prevent disability progression2. These data, integrated among a synthesis of other studies, led to the viewpoint that numerous mechanisms beyond plaques, similarly called “smoldering MS”, contribute to disease progression3. Indeed, smoldering is not restricted to inflammation in MS, but also encompasses other pathological changes including axon demyelination and mitochondria dysfunction, among others3. The smoldering model does not dismiss plaques as irrelevant, but instead, recontextualizes them as one component of the combined pathogenic dysfunctional process responsible for MS progression. Similarly, in AD, we recognize that the toxic processes that drive disease progression extend far beyond the focal, clinically measurable event of Aβ plaque deposition, and that a complex constellation of pathological changes is at play. This interplay of pathologies, including soluble Aβ (oligomers and protofibrils), insoluble Aβ (plaque), tau, inflammation, and neurodegeneration, all contribute to smoldering AD.
Before the smoke appears
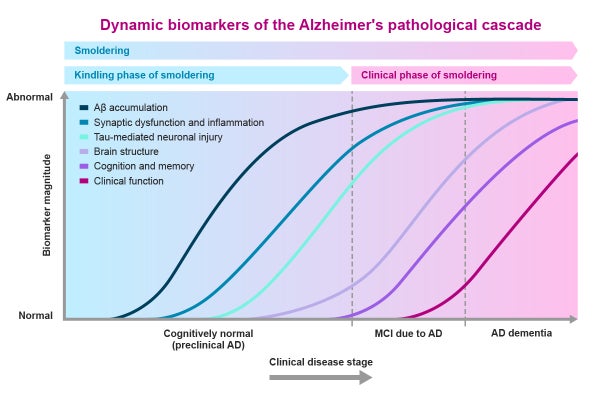
Decades before the onset of clinical symptoms in AD, a silent accumulation of neurotoxic material – referred to as “kindling” – sets the stage for smoldering disease activity. In the healthy brain, single units of Aβ, called monomers, are involved in several normal physiological functions, including processes required for learning and memory, among others4. In AD, normal Aβ production and clearance mechanisms are impaired, which starts the kindling process and causes these once-healthy Aβ monomers to misfold and aggregate into larger toxic Aβ structures called oligomers and protofibrils4. The oligomers and protofibrils diffuse throughout the brain and cause further pathological changes, including synaptic dysfunction, inflammation, and tau accumulation into toxic NFTs4,5. To counteract the kindling process, oligomers and protofibrils are sequestered into Aβ plaques, which also exert their own local toxicity4. This relentless cascade can occur unnoticed in a seemingly healthy person for up to 20–30 years4,6 until the brain’s neural reserve is ultimately overwhelmed, igniting the onset of clinical symptoms (Fig. 1).
Clinical symptoms and smoldering Alzheimer’s disease
While certain pathologies of smoldering AD (for example, tau) correlate more strongly with clinical decline, clinical symptoms are a consequence of collective brain injury driven by all components of smoldering AD, including toxic oligomers, protofibrils, plaque, inflammation, and neurodegeneration (Fig. 2). The complex interplay of smoldering pathology leads to the appearance of initially subtle clinical symptoms, which progress to mild cognitive impairment, and then mild, moderate, and eventually severe dementia. Recognizing and diagnosing AD early is crucial, since the currently available anti-amyloid therapies benefit patients in the early stages.
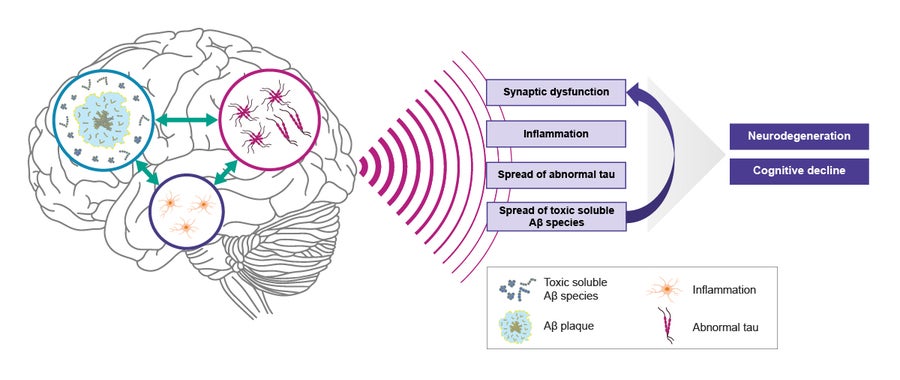
Figure 2. Proposed mechanisms of smoldering Alzheimer’s disease. Abnormal Aβ, abnormal tau, and inflammation are key pathological processes that result in the progressive neurodegeneration and cognitive decline characteristic of AD. Soluble Aβ species are particularly important in fueling smoldering processes, in part due to their ability to diffuse readily throughout the brain, versus the immobile insoluble species. The extent and rate of clinical progression can vary considerably from person to person. Aβ, amyloid beta; AD, Alzheimer’s disease.
Figure developed by Eisai Inc.
Researchers are actively working to understand what happens to smoldering disease processes and resulting clinical symptoms if one or more forms of Aβ are removed with an anti-amyloid therapy. In the revised criteria for diagnosis and staging of AD by the Alzheimer’s Association (AA) workgroup, researchers noted that while clinical trials of anti-amyloid therapies support a positive relationship between the reduction of insoluble Aβ (plaque) on positron emission tomography (PET) imaging and clinical outcomes, a reduction of insoluble Aβ below detectable thresholds does not mean that the underlying pathogenic disease processes have all been stopped1. Indeed, after stopping treatment with an anti-amyloid therapy, patients have shown re-accumulation of pathological biomarkers and a reversion to placebo rates of clinical decline1,7. The AA workgroup introduced the term “treatment-related amyloid clearance” (TRAC) for patients treated with an anti-amyloid therapy who have changed from positive to negative on amyloid PET, yet in whom the pathogenic process is still active1. This term aligns with the concept of smoldering disease, in that even if Aβ plaque is reduced to negative levels on imaging, soluble forms of Aβ, including protofibrils, as well as other aspects of smoldering AD including tau and inflammation, continue to drive disease progression.
Implications for clinical practice
The concept of smoldering AD aligns with other chronic diseases, where progress has been made in understanding the multifaceted nature of smoldering disease to develop effective treatments targeting multiple pathologies. This refreshed model of AD pathophysiology arrives at a time when significant gains have been made in the field. Over the past several years there has been a cascade of global regulatory approvals for the two available anti-amyloid therapies for patients with early AD, one directed against aggregated soluble (oligomers and protofibrils) and insoluble Aβ, and the other directed against insoluble Aβ alone8. As we now understand, plaque removal alone does not address other aspects of smoldering pathology that continue to drive clinical deterioration in its absence. Smoldering AD helps to conceptualize the importance of intervention beyond Aβ plaque clearance, including the ongoing removal of toxic soluble Aβ species that fuel ongoing smoldering disease. Furthermore, targeting synaptic dysfunction, inflammation, and tau are important considerations to effectively address smoldering disease in AD.
Finally, the kindling phase of smoldering AD reinforces the need for early diagnosis and intervention before clinical symptoms appear. Currently, anti-amyloid therapies are approved for use in early AD patients experiencing clinical symptoms. However, we know that silent damage due to kindling processes occurs for decades prior to clinical decline. While implementation of a preclinical diagnostic approach requires ethical considerations as well as appropriate identification of individuals at high risk of progressing to symptomatic AD, the field is heading in this direction, with multiple trials investigating the use of anti-amyloid therapies in individuals with preclinical AD underway. Successful trials may someday lead to a world without symptomatic Alzheimer’s disease.
Through the concept of smoldering AD, we hope to broaden disease state discussions beyond just plaque, to consider the chronic and truly multifaceted nature of this devastating disease. Our vision is to support the effort of a timely diagnosis and early treatment, to provide the best care for patients in need.