The world is experiencing a hidden epidemic with kidney disease. More than 850 million people worldwide have some form of kidney disease, which is roughly double the number of people who live with diabetes and 20 times higher than the prevalence of cancer worldwide or people living with HIV/AIDS. Furthermore, between 4.9 million and 9.7 million people worldwide require dialysis or transplantation, yet many do not receive these treatments because of lack of resources or financial barriers.
The outlook for many patients with chronic kidney disease (CKD), including its relatively rarer immune-mediated and genetic forms, has continued to be bleak. CKD leads to progressive loss of kidney function, often requiring dialysis or kidney transplantation—both of which carry substantial risk and complications—and may even result in early death.
Fortunately, a wave of new research and development is underway for serious CKD through approaches that target underlying drivers of disease.
“We’re living through a renaissance in renal medicine—a moment where deep understanding of biology and cutting-edge technology are converging to potentially change outcomes for patients,” says Carmen Bozic, chief medical officer of Vertex Pharmaceuticals. Driven by this scientific momentum, Vertex is among the companies pioneering and investigating pathway-specific approaches that are rooted in understanding and addressing the biological causes of kidney disease.
Vertex scientists are applying insights from biochemistry and genetics to explore new pathways for targeted therapies that could transform the outlook for people living with serious kidney diseases. Vertex’s research highlights how advancing the understanding of immune-mediated and genetic forms of kidney disease could one day redefine how these complex conditions are treated, based on deep understanding of their origin and underlying causes.
LIFE WITH IgA NEPHROPATHY
One of the clearest examples of this renaissance is in IgA nephropathy (IgAN), a serious B-cell-mediated kidney disease that can disrupt nearly every part of a person’s life. People living with IgAN usually have a progressive and initially asymptomatic disease until it advances, when they may experience symptoms including severe nausea, swelling, fatigue and even difficulty breathing. But the emotional toll can be even more profound. Because the disease can progress silently for years, patients may feel like they are living with an invisible threat with no real options to address the cause.
Maureen Sackett, a certified nursing assistant and patient advocate who has been living with IgAN since her diagnosis at the age of 27, recalls how her own journey began: “When I first noticed symptoms, and first talked to a physician, there was no urgency. I wasn’t told, ‘You need to see the nephrologist now. You may have kidney disease.’ At the time, with the stress of being a single mom, a nursing student, working full-time in the hospital, I was basically in survival mode at that point—so I just put it off.”
But since Maureen’s diagnosis, the IgAN landscape has evolved. Emerging discoveries are clarifying the complex biology of IgAN and renewing the drive, among clinicians and scientists alike, to act earlier and change the course of disease.
UNDERSTANDING IgA NEPHROPATHY
IgAN damages the tiny blood vessel networks (glomeruli) responsible for filtering blood in the kidneys. IgAN can occur at any age, but it most commonly affects people in the prime of their life, between 20 and 40 years old.
Although IgAN often advances slowly, up to 72 percent of patients progress to end-stage kidney disease within 20 years of diagnosis. There are insufficient treatments addressing upstream drivers of disease and therefore an unmet need remains.
At its root, IgAN is one of a number of immune-mediated diseases driven by uncontrolled B cells, where the immune system isn’t doing its job correctly. When B cells receive the wrong signals, those B cells produce abnormal antibodies that ultimately go on to mistakenly attack healthy kidney tissue, causing inflammation and injury.

IgAN has a serious impact on those affected.
Vertex Pharmaceuticals
THE ROLE OF B-CELL BIOLOGY IN IgA NEPHROPATHY
In recent years, scientists have increasingly understood that two key signaling molecules—small proteins referred to as cytokines—called BAFF (B-cell-activating factor) and APRIL (a proliferation-inducing ligand), play fundamental, overlapping but nonredundant roles in the development, differentiation, function and survival of B cells by binding to receptors on the surface of B cells.
BAFF and APRIL levels are elevated in people living with IgAN, as well as in other immune-mediated diseases, including primary membranous nephropathy (pMN), lupus nephritis and generalized myasthenia gravis. In people living with IgAN, elevated BAFF and APRIL levels drive excess production of galactose-deficient immunoglobulin A1 (Gd-IgA1) as well as disease-causing antibodies that mistakenly recognize Gd-IgA1 as a foreign substance. These self-reactive antibodies, or autoantibodies, bind Gd-IgA1 to form damaging immune complexes that accumulate in the kidney’s glomeruli. This triggers inflammation and progressive injury. Multiple studies have shown that when you reduce the levels of Gd-IgA1, there is a corresponding reduction in protein in the urine (proteinuria). A study from the Registry of Rare Kidney Diseases (RaDaR) demonstrated there is a worsening of outcomes directly correlated to the amount of proteinuria. Proteinuria reduction therefore has been an accepted surrogate endpoint predicting kidney survival outcomes. This suggests that reducing proteinuria has the potential to reduce the rate of decline in kidney function—measured as the estimated glomerular filtration rate (eGFR)—in this disease.
BAFF plays critical roles early in B-cell development but is also needed as B cells move into their middle age, while APRIL begins to act later along the B-cell development pathway, playing an important role in the maturation and survival of antibody-producing plasma cells. Given their overlapping but nonredundant roles in B-cell maturation, there is a need to inhibit both BAFF and APRIL to optimally target the causal biology that drives B-cell-mediated diseases.
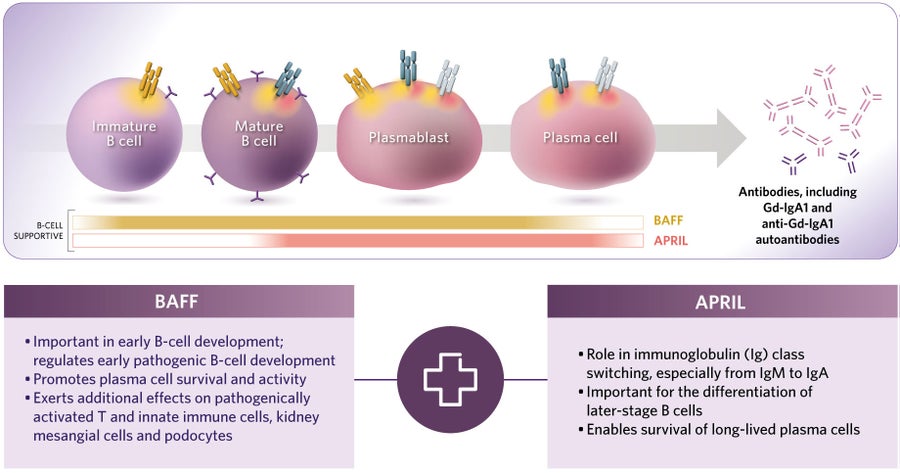
Two cytokines, BAFF and APRIL, play important roles in B-cell development.
Vertex Pharmaceuticals
CLINICAL MARKERS OF UNCONTROLLED B CELLS IN IgAN
Laboratory markers used to monitor disease progression are:
Proteinuria—excess protein in urine, a sign of ongoing kidney damage.
Hematuria—blood in urine, signaling inflammation and glomerular injury.
Circulating Gd-IgA1—elevated levels of the abnormal antibody that initiates the disease.
eGFR—a measure of kidney function that declines more rapidly as inflammation and scarring progress.
Recognizing the role of these clinical markers, researchers across the scientific community are pursuing a strategy aimed at advancing more targeted approaches for IgAN—a goal shared by experts in the field.
“I am excited about the multiple investigational treatments in development for IgAN. These therapies could move us from supportive care to addressing the key drivers of disease. Achieving sustained control of B-cell-mediated disease activity should mitigate disease progression in IgAN,” notes Brad Rovin, the Lee A. Hebert Distinguished Professor of Nephrology at the Ohio State University Wexner Medical Center.
Vertex published results of an investigational phase 1/2 trial to examine the effects of dual inhibition of BAFF and APRIL in IgAN and pMN (NCT05732402). This research is currently in a phase 3 trial in IgAN (NCT06564142) and a phase 2/3 trial in pMN (NCT07204275).
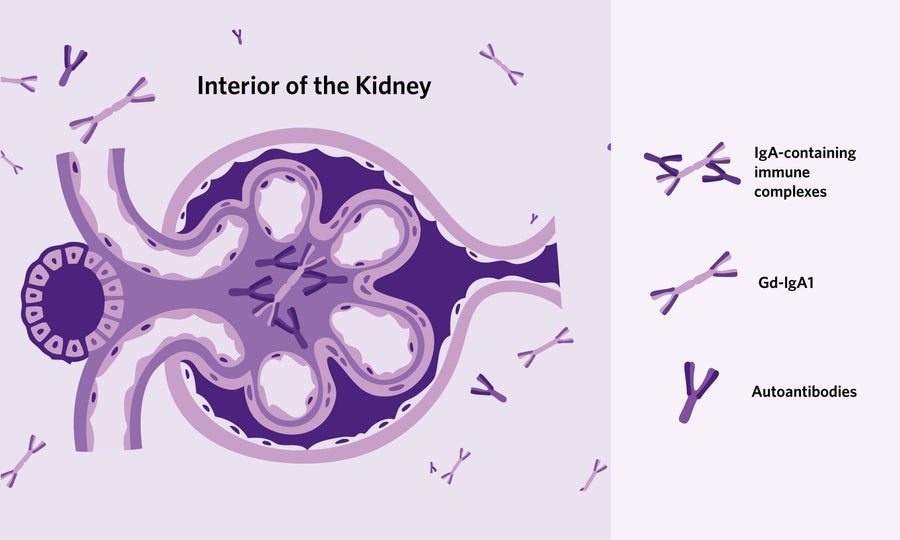
Immune complexes resulting from abnormal IgA deposit in the kidneys, triggering damage and inflammation.
Vertex Pharmaceuticals
THE HOPE FOR GENETIC KIDNEY DISEASES
New findings in immune-mediated kidney diseases are not the only insights driving this renal renaissance. Researchers are also making strides in genetic forms of CKD, where inherited mutations can predispose individuals to severe and progressive conditions.
“For the first time, medical researchers across our industry are not just treating the manifestations of genetic kidney disease—we’re studying its underlying causes and origins,” says Ogo Egbuna, vice president of clinical development of Vertex.
A compelling example of progress against genetic forms of CKD is APOL1-mediated kidney disease (AMKD), which predominantly affects people of African ancestry. In 2010, scientists discovered that individuals who inherit two variants of the APOL1 gene are at a significantly increased risk of developing kidney disease. That insight has spurred a wave of discovery aimed at inventing new investigational medicines targeting AMKD.
This deeper genetic understanding doesn’t remain in the lab; it also directly shapes the experiences of people navigating CKD.
“After a year of not having any clarity, I found out I had the APOL1 genetic mutation, and I was elated to finally have an answer to so many questions. It was a relief to know what was causing my symptoms,” says Emani McConnell-Brent, who was first diagnosed with complications from AMKD in her senior year of high school. “Increased use of genetic testing can bring hope to patients today, so that they don’t have to face the same uncertainty I did.”
Many people diagnosed with CKD may not realize their disease could be genetic; oftentimes, many people in the same family may be afflicted, and finding out the genetic origin of their disease can open new avenues for clinical trial participation and other treatment options.
A FUTURE BUILT ON BIOLOGY
The scientific community is working to transform outcomes for IgAN, genetic kidney disease and other serious conditions by addressing the underlying cause of disease. Just as past decades saw revolutions in oncology and rare genetic disorders, the next great wave of medical progress may come from nephrology as scientists, physicians and researchers are excited by emerging insights on how to manage decline and restore balance in the immune and cellular systems.
“We are at a pivotal moment in kidney disease research,” says Mark Bunnage, chief scientific officer of Vertex. “Each new discovery brings us closer to a future where treatments could address the underlying cause of these diseases and patients can also have more options.”
To learn more about Vertex’s research, visit vrtx.com.



