Key concepts
Chemistry
Electricity
Battery
Chemical reaction
Introduction
Have you ever wondered exactly how your phone, laptop or a flashlight manages to work without being plugged into a power outlet? Where does the electrical energy come from that makes all these portable devices function? You probably know the answer: They use batteries! But do you know how these batteries work? Batteries store electrical energy in the form of chemical energy, which means that electrochemical reactions inside the batteries create electricity. It may sound complicated but it is simpler than you think! In this activity you will create a basic homemade battery with just construction paper, vinegar, salt and a handful of pennies and washers—and prove it works by lighting an LED!
Background
Electricity is the presence of an electric charge, which can be either positive or negative. An electric current is generated by moving charges, usually in the form of electrons or ions. In batteries these moving charges are created from chemical reactions, meaning electrical energy is derived from chemical energy. The main components of a battery are two electrodes, typically made from carbon or two different metals, and the electrolyte, which is a liquid or paste that is in contact with both electrodes. The electrodes and electrolyte need to be electrically conductive to allow electrons and ions to flow from one electrode to the other. The question now is where do the electrons come from? Here is where chemistry comes into play.
On supporting science journalism
If you're enjoying this article, consider supporting our award-winning journalism by subscribing. By purchasing a subscription you are helping to ensure the future of impactful stories about the discoveries and ideas shaping our world today.
Electrons are generated via electrochemical redox (reduction–oxidation) reactions, in which negative charges (in the form of electrons) are transferred from one chemical (or metal) to the other. The electrons and ions released during these reactions travel through the electrolyte from one electrode to the other. During that process one electrode releases electrons while the other accepts them to complete the electric circuit. There are many different battery types that use different chemical reactions to generate electrons; two common ones are lithium ion and nickel–cadmium. In this activity you will make a copper–zinc battery using a vinegar and salt solution as the electrolyte. Do you think your battery will generate enough electricity to power an LED?
Materials
20 metal washers (galvanized; the size of a penny)
20 pennies (preferably shiny)
Pencil
Construction paper
Scissors
Bowl
Vinegar
Table salt
Spoon
Small white or red LED
Paper towels
Work area that can tolerate vinegar spills
Calculator (optional)
Aluminum foil (optional)
Multimeter (optional)
Preparation
Note: In this activity you will make a very low-voltage battery. The amount of electricity generated by this homemade battery is safe; higher voltages of electricity, however, can be very dangerous and even deadly, and you should never experiment with commercial batteries or wall outlets.
Trace a coin 20 times on the construction paper with a pencil.
Cut out all the coin-size paper pieces.
Pour some vinegar into a bowl and add enough salt to create a saturated solution, which means not all of the salt is able to dissolve. Mix with a spoon.
Put 15 of the coin-size paper pieces in the bowl and let them soak in the vinegar–salt solution for five minutes.
Procedure
Take one washer and place it on your work area. What material is the washer made of?
Take a soaked construction paper piece and place it on top of the washer. Why do you think the construction paper needs to be soaked in the vinegar–salt solution?
Next place another washer on top of the soaked paper piece.
Then place another washer on top of that washer. Add another piece of soaked construction paper and then add two more washers on top of that.
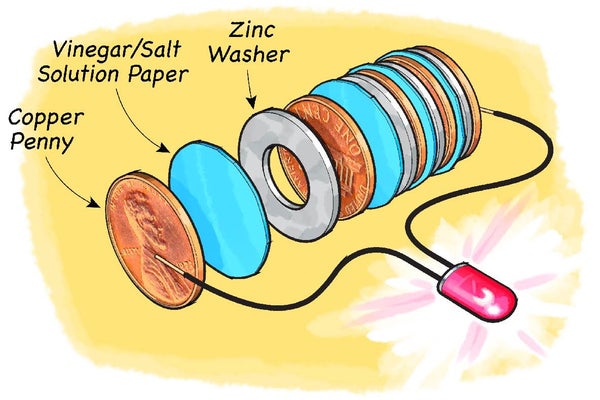
Repeat alternating the soaked paper and two washers until you have used nine washers in total. You should finish with two washers on top of a soaked piece of paper.
With a paper towel dry off the sides of your washer stack. You want to make sure it is dry on the side.
Also, check that the soaked paper is not touching more than one washer on each side.
Take the LED and spread the two contact pins apart. Then push the long pin of the LED underneath the stack so that it makes firm contact with the washer on the bottom. Place the short pin on top of the washer on the stack and press it down. Watch the LED. Does the light turn on when you connect the pins to the top and bottom of the washer pile?
Make a second stack in the same way, but this time use pennies instead of washers. What material are the coins made of?
Once the penny stack is complete, dry it off on the sides and ensure the soaked paper only touches one penny on each side.
Then take the LED again and connect the long pin to the bottom coin and the short pin to the top one. Do you see the LED light up when you make contact with the coins?
Make a third stack, but this time start with a penny on the bottom, place a soaked paper piece on top of the coin and then add a washer on top of the paper. Repeat, adding a coin, soaked paper and washer, until you have used five coins in total. You should end up with a coin placed on top of a washer.
Again, make sure to dry any excess liquid from the soaked paper on the side of the coin–washer stack, and check that the soaked paper only touches one washer and coin on each side.
Then connect the long end of the LED pins to the coin at the bottom of the pile and the short end to the washer on top of the stack. What happens to the LED this time?
Finally, use the dry construction paper pieces and make a fourth stack, alternating coin, a dry paper piece, washer, coin, a dry paper piece, washer until you have used five coins. Do you think it makes a difference if the construction paper is wet or dry?
Take the LED one more time and connect the long pin to the bottom of the stack and the short pin to the top. Does the LED light up this time? Why or why not?
Extra:How many coins and washers do you need to light up the LED? Does the amount matter at all? Try lowering the number of coins and washers you stack. What is the minimum number of coins and washers you need to light up the LED?
Extra: What would happen if you exchanged the washers with another type of material such as aluminum foil?Would you still get a functional battery? Cut coin-size pieces of foil and create a stack of coins and aluminum to find out!
Extra: If you have a multimeter at home, you can measure the voltage of your battery and how much current it produces. How does the voltage and current change as you add more coins to your battery?
Observations and results
Did you manage to get the LED to light up? Probably not with the first two stacks, which consisted of only coins or washers. Pennies are coated with copper, which turns your penny into a copper electrode for this activity. The galvanized washers, on the other hand, are coated with zinc, which is a different metal and functions as a zinc electrode in your battery. The key to a functional battery is that an electrochemical reaction has to occur between the two electrodes. If both electrodes are made of the same material, no reaction will take place and no electricity will be generated.
When you alternate the pennies with the washers, however, you create a battery with two different electrodes—one zinc and the other copper. Now an electrochemical reaction can happen between the zinc and copper that releases electrons to travel through the electrolyte (vinegar and salt–soaked construction paper) to generate an electric current. This is why the LED should have lit up in the third stack that you created by alternating coins and washers. When you removed the electrolyte and used dry paper pieces instead, the electrons were no longer able to move from one electrode to the other, so no electric current was produced and the LED did not light up!
Cleanup
Rinse the coins and washers with tap water and dry them off. You can reuse them afterward. Discard the soaked construction paper and wipe down your work area.
More to explore
How Do Batteries Store and Discharge Electricity?, from Scientific American
Generate Electricity with a Lemon Battery, from Scientific American
Types of Batteries, from the Australian Academy of Science
Chemistry of Batteries, from the University of Waterloo
Science Activity for All Ages!, from Science Buddies
This activity brought to you in partnership with Science Buddies