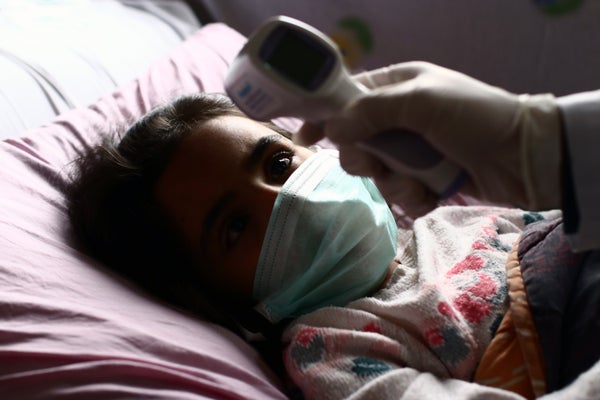
On a warm, mid-June afternoon a concerned mother brought her 11-year-old daughter, who had a high fever and a severe bellyache, to the emergency room at Hasbro Children’s Hospital in Providence, R.I. After doctors ruled out the usual suspects for the symptoms, such as bacterial infections and appendicitis, they started to seriously consider a diagnosis that would have been inconceivable two months prior: an emergent and potentially fatal inflammatory condition that occurs in children about four weeks after they are exposed to the new coronavirus.
The rare disease—called multisystem inflammatory syndrome in children (MIS-C) in the U.S. or pediatric multisystem inflammatory syndrome in the U.K., where it was first defined this past spring—is a hyperimmune response to SARS-CoV-2. It disproportionately affects Black, Hispanic and Latino children. In some cases of MIS-C resembling the bacterial overloads of toxic shock syndrome, youngsters arrive at the hospital with shock symptoms and organ failure. In others, they experience a high fever and inflammatory symptoms resembling those of Kawasaki disease, which strikes children’s blood vessels. Or the illness can look like neither of those threats, even though affected kids still have a high fever and widespread inflammation. Nearly all of the children treated for MIS-C survive and appear to be healthy afterward. But when it is undiagnosed or untreated, the condition can permanently damage the heart or cause death.
The disease is so new that there is no consensus on several of its fundamental features or on how to treat it. But insights from doctors who have been confronting MIS-C this summer, along with new research about its victims, are starting to reveal important clues. For instance, a small study published on August 18 in Nature Medicine concluded that the condition is distinct from Kawasaki disease, as many doctors suspected. And several large research efforts that have recently been launched could soon clarify how to diagnose MIS-C and identify trends in treatments that seem to work best.
On supporting science journalism
If you're enjoying this article, consider supporting our award-winning journalism by subscribing. By purchasing a subscription you are helping to ensure the future of impactful stories about the discoveries and ideas shaping our world today.
As of August 20, 694 MIS-C cases and 11 deaths had been reported to the U.S. Centers for Disease Control and Prevention. About 1,000 cases had been documented worldwide by late July. Children are hospitalized for COVID-19 more than they are for MIS-C, according to various reports, but doctors say the latter is more dangerous. Although most youngsters recover, they often do so after severe illness. According to threerecentstudies, between 64 percent and 80 percent of those hospitalized for the condition required intensive care treatment, compared with about 33 percent of children hospitalized for COVID-19.
With so few cases and descriptive studies, no diagnostic tests and no randomized controlled experimental results to guide treatment, hospital pediatricians have had to dig deep to figure out how to help the young victims. Silvia Chiang, a Brown University pediatric infectious disease (ID) specialist, and her colleague Sabina Holland managed the case at Hasbro. Keen for insight, Chiang followed evaluation guidelines developed by the Children’s Hospital of Philadelphia to the point where they read, “Consult ID”—in other words, she should consult herself. “I thought, ‘Ha ha. Okay. So now what do I do?’” she recalls.
Because there are no doctors who have any depth of experience with MIS-C, infectious disease physicians are huddling in-house with colleagues in specialties such as cardiology, hematology and rheumatology. Some of them are consulting far-flung colleagues with experience in treating the inflammatory Kawasaki disease, which has unknown causes, affects a few thousand young children annually in the U.S. and is more common in Japan.
As Chiang pursued clues for how to treat her patient, she came across limited CDC guidelines, as well as a Webinar for clinicians hosted by the agency. The latter featured a doctor affiliated with Imperial College London and St. Mary’s Hospital in London, who described dozens of area children who came down with MIS-C’s characteristic high fever and severe inflammation, which seemed to arise about a month after exposure to the new coronavirus. Chiang learned that four weeks before coming to the hospital, two of the Hasbro patient’s family members had recovered at home from relatively mild cases of COVID-19. The girl tested positive at the hospital for the coronavirus and for antibodies to it. Along with imaging scans, blood-work results and the CDC’s description, the evidence added up to MIS-C—the first reported case in Rhode Island.
Rapid, accurate diagnosis of MIS-C is crucial for treating children before the illness damages their heart or causes other life-threatening complications.“Kids come in kind of irritable and feverish and then, hours later, they just suddenly tank,” says Russell McCulloh, a pediatric infectious disease specialist at Children’s Hospital & Medical Center in Omaha. In some cases, hospitalized children quickly developed coronary artery inflammation, cardiac dysfunction such as a weakly pumping heart or even heart failure so severe that patients are placed on a so-called extracorporeal membrane oxygenation machine to do the work of the heart or lungs or on a mechanical ventilator to do the work of the latter.
U.S. children’s hospitals, the American College of Rheumatology and the American Academy of Pediatrics have recently published guidance for physicians on how to evaluate, diagnose and treat MIS-C, such as the material Chiang consulted. But that information is preliminary. As more cases advance doctors’ understanding, the guidelines will likely evolve, McCulloh says. He and his colleagues havetreated about a dozen cases now. Guidelines, however, are not as useful as diagnostic criteria, which have yet to be developed, according to pediatric rheumatologist Grant Schulert. He works with teams at Cincinnati Children’s Hospital that have treated several youngsters diagnosed with MIS-C. For now, case definitions are written so broadly, they could muddy diagnoses, Schulert says.
Another problem, he notes, is that MIS-C case definitions often specify using antibody tests for the virus, which indicate that patients have been infected at some point in the recent past. Those results are a useful marker in regions currently experiencing a peak in infections, Schulert says, but not in places in which the virus has been slowly creeping among people for many months—such as much of the Midwest, where he works. “In the winter, if we see a patient with symptoms consistent with COVID-19 who is antibody-positive, we cannot necessarily assume that they were infected in the past four weeks,” he says. “This highlights the need to understand the biology of MIS-C, so a more biologically based classification and diagnostic criteria can be constructed.”
The common thread across all cases, says Michael Levin, the pediatric infectious disease specialist who appeared in the CDC Webinar, is severe inflammation of multiple organ systems, as evidenced by blood markers, along with exposure to SARS-CoV-2. Levin’s team was the first to define MIS-C when it did so for the Royal College of Pediatrics and Child Health. The CDC and World Health Organization soon followed.
To avoid misdiagnosis, doctors have to also consider a laundry list of other conditions, ranging from tick-borne illnesses to sepsis. But the fear of missing a potential MIS-C patient can lead hospital physicians to perform excessive and unnecessary testing in patients with fevers, says pediatric infectious disease specialist Jocelyn Ang. She and her colleagues have treated at least three dozen MIS-C patients at Children’s Hospital of Michigan in Detroit.
Figuring out how to treat MIS-C is even more fraught than diagnosing it, some doctors say. “Right now we’re using really blunt treatment based off of other diseases, because we don’t have better information,” McCulloh says. Given that some MIS-C symptoms overlap with those for Kawasaki disease and other inflammatory illnesses, physicians have, with some success, relied on treatments for those conditions to tamp down the inflammation and the overactive immune system that brought it on. Treatments include intravenous immunoglobulin—an anti-inflammatory blood product—as well as steroids and monoclonal antibodies. In mild cases, some patients recover with minimal intervention that stabilizes their blood pressure or breathing while their immune system settles down. But there are no data so far to demonstrate which drugs, if any, are effective or the best course of action.
As a stopgap move until randomized controlled treatment studies are funded, researchers have launched case-registry efforts in the U.S. and U.K. In one led by Imperial College London, health care workers representing 90 different hospitals in 39 countries, including Brazil, Norway, and Pakistan, have entered their lengthy MIS-C case and treatment details in an anonymized online database. With statistical corrections for bias in treatments based on disease severity, analysts could compare how well specific approaches worked for patients across many places. With enough entries, helpful guidance could come by the end of the year, Levin says.
Similarly, the CDC has funded a study led by Boston Children’s Hospital that is collecting data at more than 70 U.S. hospitals and analyzing risk factors and outcomes among 2,000 children and young adults hospitalized with COVID-19 and MIS-C. The group reported the features and treatment of 186 patients with MIS-C in 26 U.S. states, and its analysis is ongoing, says Adrienne Randolph, asenior associate in critical care at Boston Children’s and the project’s principal investigator. And a multinational study, funded by the European Commission and led by Imperial College London, is collecting blood samples from patients with COVID-19, MIS-C and other infections. The researchers are looking for telltale compounds in blood in order to classify conditions by their molecular signature for use in diagnosis, Levin says. Initial results could come this fall.
As for Chiang’s challenging case in June, her patient recovered well and has shown no lasting problems. But a few weeks ago the girl’s mother told Chiang that her daughter was anxious, probably from the experience with MIS-C, which included two hospitalizations and multiple drug treatments. Chiang empathizes and sees the big picture. “I can imagine being a young adult like her,” she says. “You have this illness that is brand new, so doctors have very little experience in treating it. And you hear all these things in the media about how scary the disease is. I can imagine that would be a bit hard.”
Read more about the coronavirus outbreak from Scientific Americanhere. And read coverage from our international network of magazines here.