Western cultures have long cherished the notion that all people are created equal. But in the real world, our lives are not balanced with equal opportunities and resources. This distinction was noted mordantly in 1894 by author Anatole France, who wrote that “the law, in its majestic equality, forbids the rich as well as the poor to sleep under bridges, to beg in the streets, and to steal bread.” The rich, of course, need none of these things, whereas the poor often have little choice. And economic disparity has only gotten worse during the past several decades, particularly in the U.S. In 1976 the richest 1 percent of U.S. citizens owned 9 percent of the country’s wealth; today they own about 30 percent. This trend echoes around the globe.
One of the consequences for the growing poor is worsening health, and the reasons are not as obvious as you might think. Yes, lower socioeconomic status (SES) means less access to health care and living in more disease-prone neighborhoods. And, yes, as the SES ladder’s lower rungs have become more populated, the number of people with medical problems has climbed. This is not merely an issue of poor health for the poor and some version of better health for everyone else. Starting with Elon Musk at the top, every step down the ladder is associated with worse health.
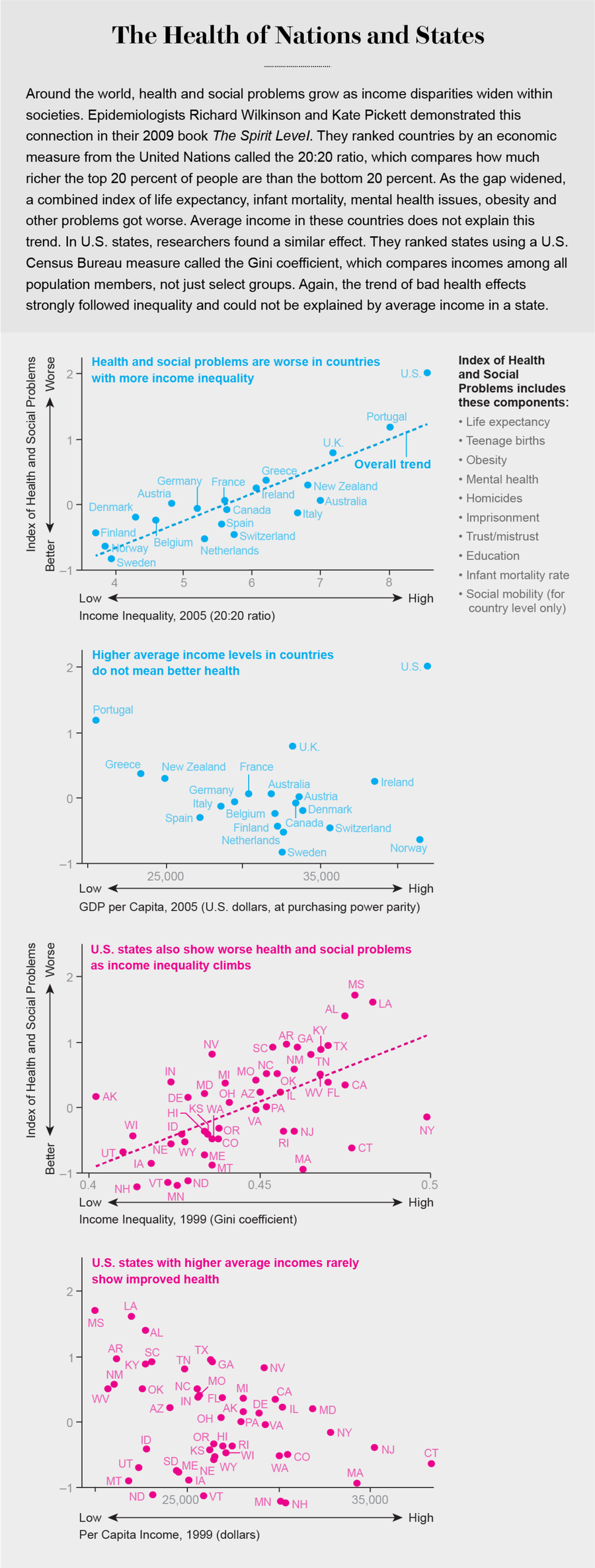
The link between socioeconomic inequality and poor health goes beyond simple access to care and living with more dangers. Fewer than half of the health changes along this SES/health ladder can be explained away by risks such as smoking, alcohol consumption and reliance on fast food or by protective factors such as insurance and health club memberships. In the U.K., large, long-term studies of risks in specific groups, led by epidemiologist Michael Marmot and called the Whitehall studies, demonstrated this clearly. Further, this ladder, or gradient, exists in nations with universal health care; if care availability were truly responsible, universal access should make the gradient vanish. Something else, something quite powerful, must be associated with inequities and be able to cause health problems.
On supporting science journalism
If you're enjoying this article, consider supporting our award-winning journalism by subscribing. By purchasing a subscription you are helping to ensure the future of impactful stories about the discoveries and ideas shaping our world today.
That factor seems to be the stressful psychosocial consequences of low SES. Psychologist Nancy Adler of the University of California, San Francisco, and her colleagues have demonstrated that how people rate how they are doing, relative to others, is at least as predictive of health or illness as are any objective measures such as actual income level. The research indicates that poor health is as much about feeling poor as being poor. Epidemiologists Richard Wilkinson and Kate Pickett of the University of Nottingham and the University of York in England, respectively, have filled out this picture in detail, showing that while poverty is bad for your health, poverty amid plenty—inequality—can be worse by just about any measure: infant mortality, overall life expectancy, obesity, murder rates, and more. Health is particularly corroded by your nose constantly being rubbed in what you do not have.
Basically, more unequal societies have worse quality of life. Across countries and among U.S. states, more inequality, independent of absolute levels of income, predicts higher rates of crime, including homicide, and higher incarceration rates. Add in higher rates of kids being bullied at schools, more teen pregnancies and lower literacy. There are more psychiatric problems, alcoholism and drug use, as well as lower levels of happiness and less social mobility. And there is less social support—a steep hierarchy is the antithesis of the equality and symmetry that nourish friendship. This grim collective picture helps to explain the immensely important fact that when inequality increases, everyone’s health suffers.
This is where the problem affects the rich, the haves, as well as the have-nots. With increasing inequality, they typically expend more resources insulating themselves from the world underneath the bridges. I have heard economist Robert Evans of the University of British Columbia call this the “secession of the wealthy.” They spend more of their own resources on gated communities, private schools, bottled water and expensive organic food. And they give lots of money to politicians who help them maintain their status. It is stressful to construct thick walls to keep everything stressful out.
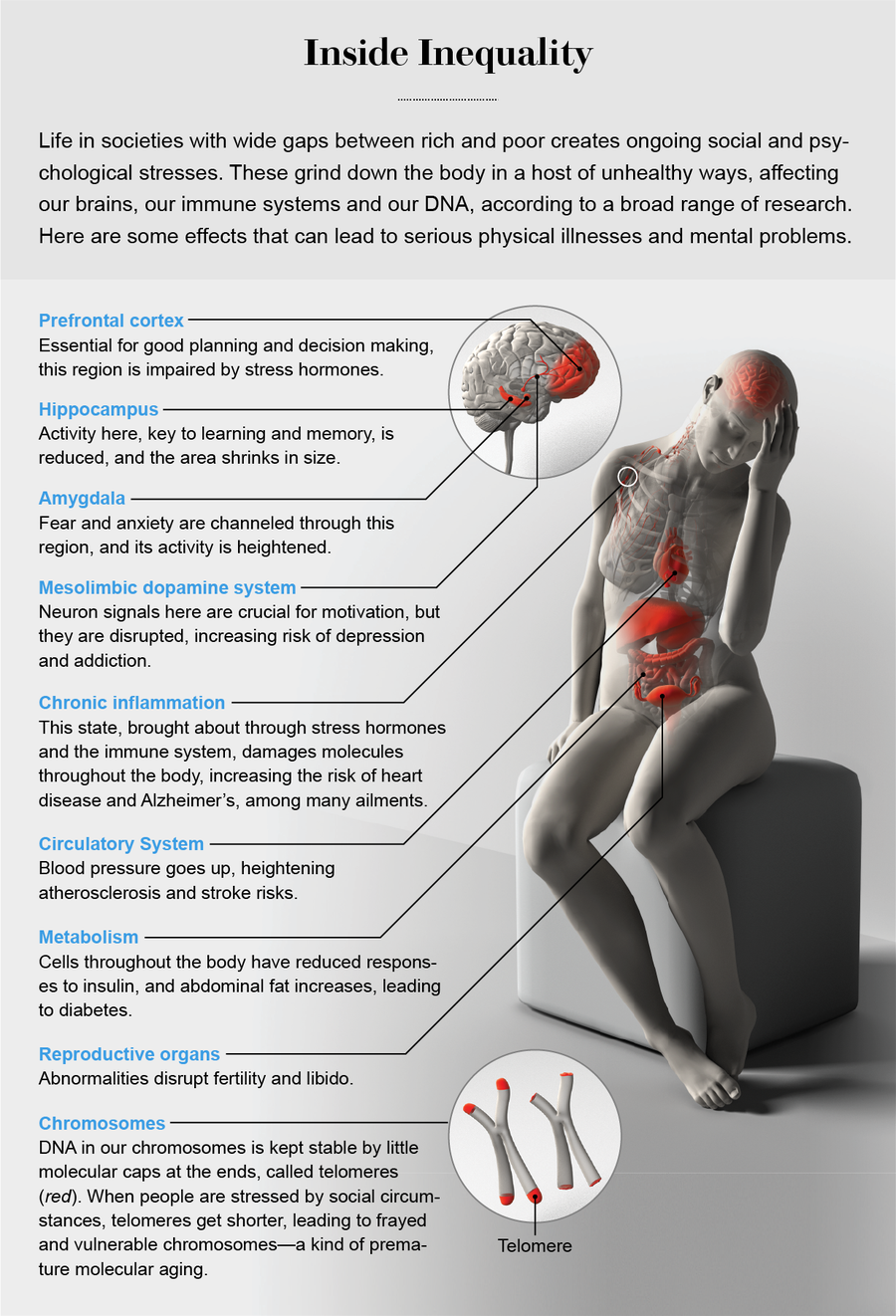
Knowing that these psychological and social factors influence the biology of disease is one thing. Demonstrating just how these stressors do their dirty work inside the body is something else. How do SES and inequality “get under the skin”? It turns out that researchers have made significant strides toward an answer. We have learned a lot about how poverty affects biology, and the part of the growing inequality gap that worries people is the poverty end. Scientists have been able to trace physiological connections from external inequality to three key inner areas: chronic inflammation, chromosomal aging and brain function.
A Heavy Load
Thinking about the biology of disease was revolutionized in the 1990s, when the late Bruce McEwen of the Rockefeller University introduced the concept of allostatic load. Our bodies are constantly challenged by our environment, and we stay healthy when we meet those challenges and return to a baseline state, or homeostasis. Traditionally this view led scientists to focus on specific organs that solve specific challenges. Allostasis has a different perspective: physiological challenges provoke far-flung adaptations throughout the body. An infected toe, for instance, will produce not only inflammation at the tip of the foot but also wider changes in everything from energy taken from abdominal fat to the brain chemistry of sleepiness. As this biological grind continues, it leads to an array of body parts functioning less than optimally, which can be as damaging to health as a single organ gone very wrong.
Teresa Seeman of the University of California, Los Angeles, took this idea and followed it through the body, measuring various biomarkers of wear and tear, including increases in blood pressure, cholesterol, blood lipids, body mass index, molecular indicators of chronic hyperglycemia, and levels of stress hormones. She showed that this group of disparate measures powerfully predicts physical health and mortality.
Recent research by Seeman and others links low SES with heavy allostatic load because the body is in a constant and futile battle to return to a normal, nonstressed state. These findings highlight an important theme: whereas an adult’s SES predicts allostatic wear and tear, childhood SES leaves a stronger lifelong mark. Low SES predisposes youngsters’ bodies toward earlier “aging.” The scientists also found protective factors. Although growing up in an impoverished neighborhood worsens the low SES–allostatic load link, lucking out with a mother who has the time and energy to be highly nurturing reduces the ill effects.
Credit: Bryan Christie Design
Stress in any form can produce these effects. It does not have to be related to money, but it is usually related to social situations. My own work with baboons living freely on the East African savanna has shown this effect. In baboon groups, an animal’s place in the social hierarchy produces more or less stress. If you are a low-ranking baboon—a socially stressful situation—your body has unhealthy abnormalities in its secretion of glucocorticoids, which are stress hormones such as cortisol. The body also shows unhealthy changes in the gonadal, cardiovascular and immune systems.
In animal and human hierarchies, these stress-induced changes affect health through a key process: chronic inflammation. Few things are better examples of a double-edged biological sword than inflammation. After tissue injury, inflammation contains damage and initiates cell repair. Chronic widespread inflammation, however, causes molecular damage throughout the body, and studies have demonstrated that it contributes to diseases ranging from atherosclerosis to Alzheimer’s. Recent work (including my own focusing on inflammation of the nervous system) indicates that chronic high stress levels can promote chronic inflammation. In people, childhood poverty upregulates the adult body’s proinflammatory set point, with increased expression of inflammatory genes and increased levels of inflammatory markers such as C-reactive protein, which is associated with a higher risk of heart attacks.
These are long-term effects: more financial losses in the Great Recession predict higher C-reactive protein levels six years later. Humans share such vulnerabilities with other primates that live in unequal circumstances. Work by Jenny Tung of Duke University shows more markers of chronic inflammation in low-ranking rhesus monkeys compared with the socially dominant animals in a group. Such studies highlight the directness of the link between social stress factors and unhealthy biology because that link occurs in a species that lacks changes in lifestyle risk factors, such as increased rates of smoking and drinking, that we often see in humans who are stuck in low-status situations.
Premature DNA Aging
Progress in understanding the routes into the body taken by the SES/health gradient has also come through a very sensitive measure of aging: the condition of telomeres, which are the stretches of DNA at the very tips of chromosomes.
Telomeres help to keep our chromosomes stable—molecular biologists like to say that they resemble the plastic caps at the ends of shoelaces that prevent fraying. Every time chromosomes are duplicated for cell division, the telomeres shorten; when they get too short, cells can no longer divide, and they lose many of their healthy functions. Telomere shortening is countered by the enzyme telomerase, which rebuilds these tips. Thus, the state of a cell’s telomeres tells much about its biological “age,” and shortened telomeres that produce frayed, vulnerable chromosomes seem to be a molecular version of wear and tear.
Telomere biology met stress physiology in a 2004 study led by health psychologist Elissa Epel and molecular biologist Elizabeth Blackburn, then both at U.C.S.F.; Blackburn won a Nobel Prize in 2009 for her pioneering work on telomeres. They examined 39 people who lived with severe stress every day: women who were caregivers for chronically ill children. The landmark finding was that white blood cells in these caregivers had shortened telomeres, decreased telomerase activity, and elevated oxidative damage to proteins and enzymes. (Oxidation can disable telomerase.) The longer a child’s illness, the more stress the women reported and the shorter their telomeres were, even after the researchers accounted for potentially confounding factors such as diet and smoking. Telomeres normally shorten at a more or less constant rate in people, and calculations showed that these women’s telomeres had aged roughly an additional decade—and sometimes more—past those in the low-stress group.
This discovery triggered a flood of supporting studies showing that stressors that include major depression, post-traumatic stress disorder and the experience of racial discrimination can all accelerate telomere shortening. Unsurprisingly, lower childhood SES also predicts shorter telomeres in adulthood; perceived poor neighborhood quality, witnessing or experiencing violence, family instability (such as divorce, death or incarceration of a parent), and other features of poor status early on are tied to these shrunken chromosome tips later in life. Spend your childhood in poverty, and by middle age your telomeres will probably be about a decade older than those of people with more fortunate childhoods.
Thus, from the macro level of entire body systems to the micro level of individual chromosomes, poverty finds a way to produce wear and tear. Most studies of telomere length compare “poor” with “non-poor,” as do the studies comparing allostatic load, but the few studies that examine the whole spectrum of inequality, step by low-status step, show that every rung down the SES ladder most likely worsens these biological markers of aging.
Out of Control
Slipping down these rungs also changes the brain and behavior, according to a slew of recent neurobiological studies. My laboratory has devoted more than a quarter of a century to studying what ongoing stress does to the brain in rodents, monkeys and humans. Along with other labs, we have learned that one hotspot is the hippocampus, a region critical to learning and memory. Sustained stress or exposure to excessive glucocorticoids impairs memory by lowering hippocampal excitability, retracting connections between neurons and suppressing the birth of new neurons. In the amygdala, a brain area that is central to fear and anxiety, stress and glucocorticoids heighten those two reactions. Instead of damping things down as they do in the hippocampus, in this fear-promoting region they increase excitability and expand neuronal connections. Together these findings help to explain why post-traumatic stress disorder atrophies the hippocampus and enlarges the amygdala. Another affected area is the mesolimbic dopamine system, which is crucial to reward, anticipation and motivation. Chronic stress disrupts that system, and the result is a predisposition toward the anhedonia of depression and vulnerability to addiction.
Bombardment by glucocorticoids also affects the prefrontal cortex (PFC), key to long-term planning, executive function and impulse control. In the PFC, social stress and elevated glucocorticoids weaken connections between neurons, making it harder for them to communicate. Myelination, the process that insulates cables between neurons and thus helps them pass signals faster, is impaired. Total cell volume in the region declines, and chronic inflammation is activated.
Credit: Jen Christiansen; Sources: The Spirit Level: Why Greater Equality Makes Societies Stronger. Kate Pickett and Richard Wilkinson. Bloomsbury, 2009; Bureau of Economic Analysis, U.S. Department of Commerce (U.S. income per capita data); Human Development Report 2007/2008: Fighting Climate Change: Human Solidarity in a Divided World. United Nations Development Program, 2007 (GDP per capita data)
What happens when the PFC is impaired in this way? Lousy, impulsive decisions happen. Consider “temporal discounting”: when choosing between an immediate reward and a bigger one if you wait, the appeal of waiting goes down as the time you have to wait goes up. The PFC is normally good at combating this shortsightedness. But stress steepens temporal discounting; the more cumulative stress, the less PFC activation in experiments that call for gratification postponement. For people sliding further into inequality, the less active PFC makes it harder for the brain to choose long-term health over immediate pleasure. That neurological effect might explain why people with more total life stress gain more weight and smoke and drink more than those with fewer stressors.
These changes in the PFC happen in children, too. In separate studies, Martha Farah of the University of Pennsylvania and W. Thomas Boyce, professor emeritus at U.C.S.F., observed that lower-SES kindergartners typically have elevated glucocorticoid levels, a thinner and less active PFC, and poor PFC-dependent impulse control and executive function. These effects increase as kids get older. By adolescence, lower SES predicts smaller PFC volume. By adulthood, low SES predicts steeper temporally discounted decisions.
Some of these observations present a tricky chicken-and-egg question. The brain changes could lead to poor choices, which in turn lead to deeper poverty, rather than the other way around. But the research suggests that causes and effects run in the other direction, with SES and inequality first influencing PFC function and then other bad things happening.
For example, kindergartners’ SES predicted their PFC function; few five-year-olds plummet into poverty by squandering their paychecks on drink and horses. Further evidence comes from a 2013 study by Jiaying Zhao of the University of British Columbia and her colleagues. They examined Indian farmers whose economic fortunes vary seasonably. As individuals’ SES went from being poorest during planting season to wealthiest after harvest, improvements in PFC function followed.
To me, the most important evidence comes from research in which people’s sense of their SES was lowered by the design of the experiment. Afterward these individuals did heavier temporal discounting. In one 2012 study, subjects played a game of chance against one another, with differing amounts of starting resources. “Poor” subjects became more likely to borrow against future earnings and less attuned to helpful clues about game strategy.
In another study, subjects who were prompted to imagine scenarios of financial loss (versus neutral or advantageous ones) did steeper temporal discounting in an unrelated task. In still other research, subjects were primed to imagine their financial burdens by contemplating an expensive car repair; cognitive function was unchanged in high-SES subjects but declined in poorer individuals.
Why should a transient sense of lower SES induce cognitive changes typical of lower SES in the real world? One explanation is that it is a rational response because it is hard to think about squirreling away money for old age if you can barely buy groceries. Poverty makes the future a less relevant place.
But there is also a powerful stress-related explanation: long-term planning and impulse control tires out the PFC. Increase subjects’ cognitive “load” with taxing PFC-dependent tasks, and they become more likely to cheat on their diet. Or you can—and scientists have done this—increase cognitive load by tempting dieting subjects with snacks, and then they do worse on PFC-dependent tests. How much this represents literal “depletion” of the PFC metabolically versus declining motivation is unclear.
Either way, lower SES creates chronic financial worry that distracts and exhausts. It is hard to ace a psychological task of, say, subtracting a series of numbers or a more important task of reining in your drinking when you are worrying about paying your rent. One finding in the car-repair study supports this interpretation. When subjects contemplated a repair of negligible cost, low- and high-SES subjects performed equally well on cognitive tasks.
Of course, we need to better understand the biological consequences of inequality and learn better ways to heal its health scars. But frankly, right now we know quite a bit. We know enough to prompt moral outrage at the situation. It is outrageous that if children are born into a disadvantaged family, they will be predisposed toward poor health by the time they start to learn the alphabet. It should not require us to measure inflammation or the length of chromosomes to prove this is wrong, but if it does, more power to this science.

