Editor’s Note (6/23/23): On June 22 the U.S. Coast Guard announced that a remotely operated vehicle found debris from the Titan submersible. OceanGate Expeditions, the company that owns the vehicle, declared that the Titan and all five people onboard were lost. For more coverage of the submersible, see “What Happened to Imploded Titanic Tourist Sub?”
Search-and-rescue efforts continue for the Titan submersible, an underwater craft that went missing on Sunday with five people onboard during a dive to the shipwrecked remains of the Titanic. The submersible does not seem to be at the water’s surface, because attempts to locate the craft from the air have searched 10,000 square miles of ocean without a glimpse. Sonar buoys did, however, detect underwater banging noises within the search region. Remotely operated diving vehicles have been targeting the location of those sounds.
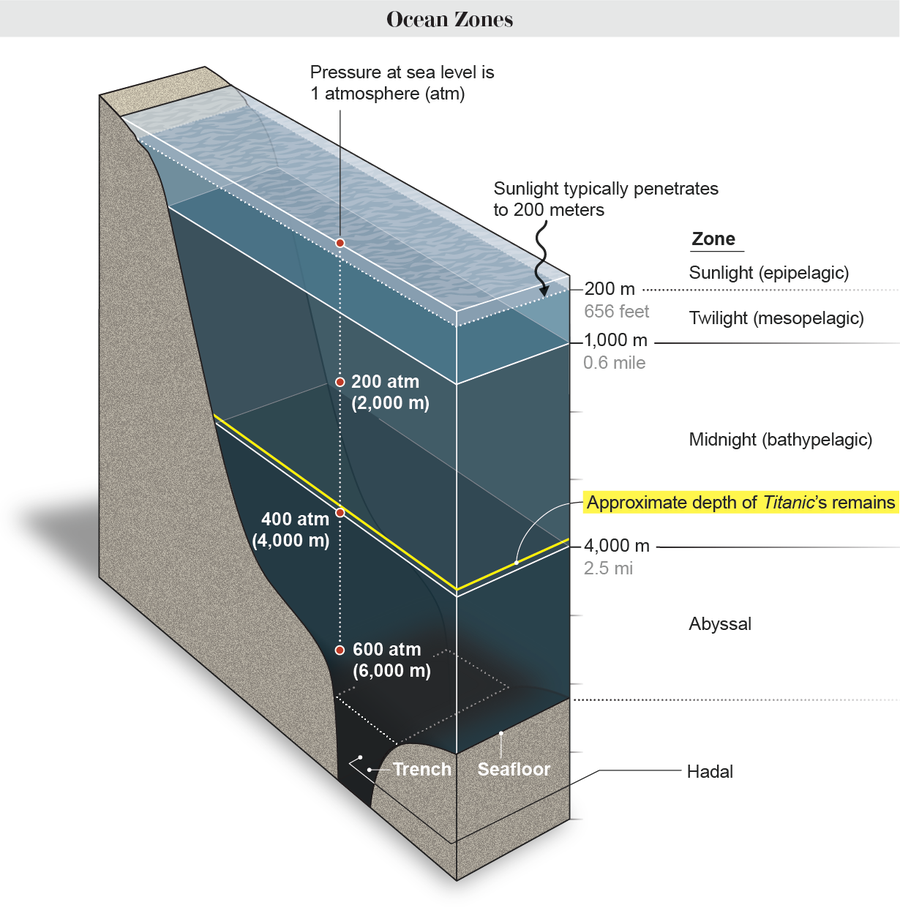
If the Titan has become stranded at lower depths, one of the greatest dangers it faces is increasing water pressure as the ocean gets deeper. Everything on Earth’s surface experiences some pressure from the weight of the air above us. That’s why the pressure at sea level is often characterized as one atmosphere, a unit equivalent to almost 15 pounds per square inch (psi). Start descending into the ocean, and the air’s weight is joined by that of the water. For every 33 feet (10 meters) of saltwater depth, pressure increases by another atmosphere.
Credit: Jen Christiansen, modified from “How the Ocean Sustains Complex Life,” by Mark Fischetti, Kelly J. Benoit-Bird, Skye Morét and Jen Christiansen, in Scientific American; August 2022
Scientists have divided the ocean’s depths into various zones, and the light and pressure in each one determines what kind of life can survive there. For instance, a variety of plants and animals thrive in the epipelagic, or sunlight, zone—the top layer of water, where light is strong and pressure is relatively low. But sunlight typically only penetrates water to a depth of 656 feet, where the water pressure is about 21 atmospheres, or roughly equivalent to two adult humans balancing on a one-square-inch spot.
To reach the remains of the Titanic, a vessel must go much lower than that. The wreck sits in the dark bathypelagic, or midnight, zone, at a spot 12,400 feet below the ocean’s surface. The pressure around the famous sunken ship is about 375 atmospheres. That means every square inch of an object’s surface experiences the equivalent of 5,500 pounds of force. Such an amount is greater than the bite pressure exerted by some of the strongest jaws in the animal kingdom: Crocodile bites have been measured at up to 3,700 psi. And one computer modeling study has estimated that a relatively large great white shark could sink its teeth into prey with a force of almost 4,000 psi.
For submersibles to survive the equivalent of being crushed in a great white shark’s jaws, they must be built of strong materials and have a shape that will withstand the water pressing in on all sides. To cope with the latter, many of these vehicles are spherical; the missing Titan is cylindrical. On top of that, a deep-sea submersibles require an oxygen supply for their passengers, as well as carbon dioxide scrubbers to prevent those passengers from suffocating in their own exhaled breath, a heating system to withstand the cold at depths sunlight never reaches, and sensing and navigation systems to guide the vessels as they descend, locate a target such as the shipwreck of the Titanic—and return to the surface.

