Each decade seems to have its signature environmental crisis that dominates headlines. Over time those issues seemingly disappear, leaving us to idly wonder from time to time if problems like the ozone hole or acid rain are still planetary concerns or if we’ve solved them.
On this Earth Day, Scientific American is here to give you a status update on some of the major environmental stories of past decades.
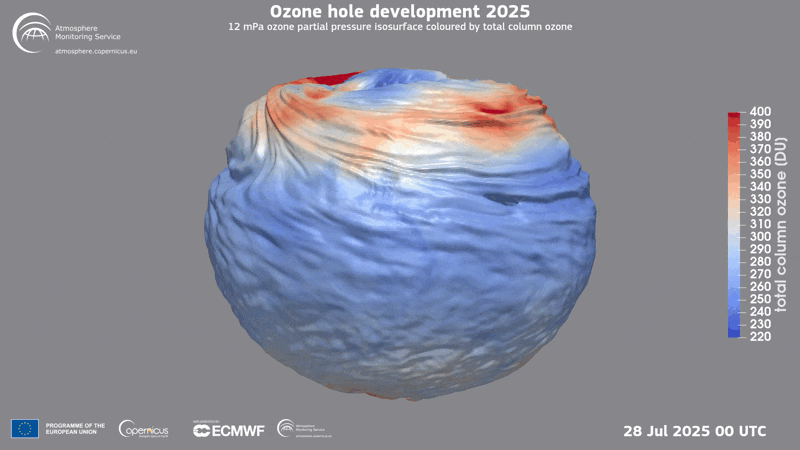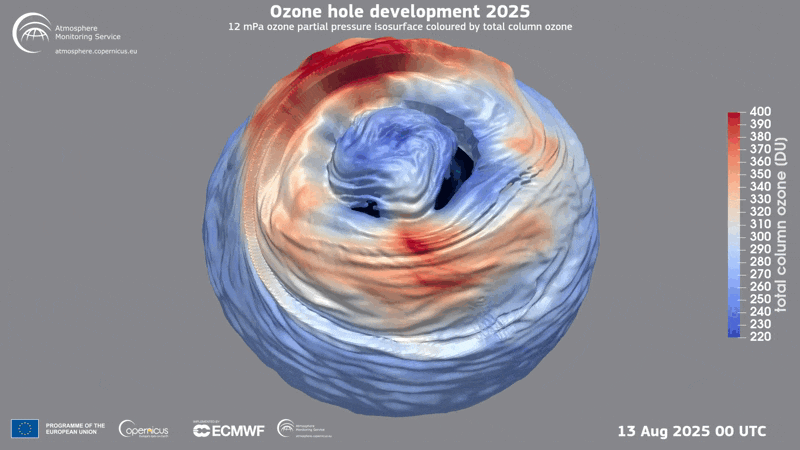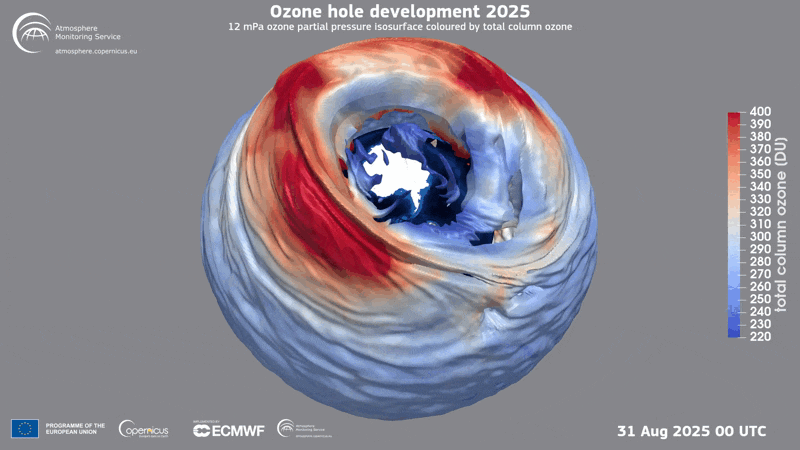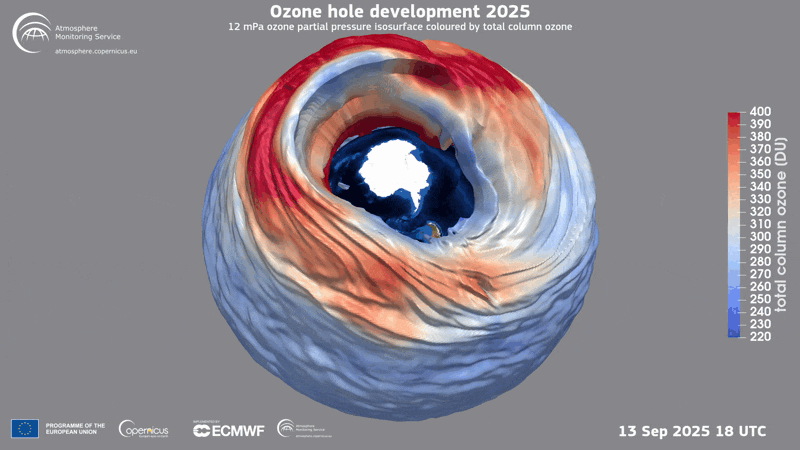
A three-dimensional rendering of the ozone hole’s evolution in 2025.
CAMS
On supporting science journalism
If you're enjoying this article, consider supporting our award-winning journalism by subscribing. By purchasing a subscription you are helping to ensure the future of impactful stories about the discoveries and ideas shaping our world today.
Ozone Hole
In 1985 three British scientists announced their discovery of a massive hole in Earth’s protective ozone layer that was opening every spring over Antarctica. The culprits, it turned out, were chlorofluorocarbons (CFCs) and certain other chemicals used in refrigeration and aerosol sprays. (Another trio of scientists had warned the world about CFCs in the 1970s and later won the Nobel Prize in Chemistry for it.) When CFCs break down in the atmosphere, they release chlorine that leads to the destruction of ozone, a molecule that contains three oxygen molecules and blocks harmful ultraviolet rays from reaching the planet’s surface.
As people became alarmed about the possibility of rising skin cancer rates, nations met in 1987 to pass the Montreal Protocol and phase out CFCs. It was the first United Nations treaty to be universally ratified and was possible in part because of ready alternative chemicals.
Threats to the ozone layer continue, from, for example, chlorine-containing molecules emitted by massive wildfires and aluminum oxide produced when satellites and other space objects burn up upon reentry into the atmosphere. Even so, CFC levels have plummeted since the Protocol was enacted, and the ozone layer has begun a long, slow recovery.
Current predictions suggest the ozone layer will recover to its state in 1980 over the tropics and middle latitudes by 2040, and it will reach that point over the Arctic by about 2045 and over Antarctica by the mid-2060s.
“The Montreal Protocol is the best environmental agreement we’ve ever created,” Durwood Zaelke, an environmental policy expert at the University of California, Santa Barbara, and founder of an organization focused on reducing climate pollutants, told Scientific American in 2025.

Acid rain damage to the entrance of the Church of the Holy Cross in Coimbra, Portugal.
jacquesvandinteren/Getty Images
Acid Rain
In the 1980s and 1990s one of the top environmental crises grabbing public attention was acid rain, with news reports showing images of the partially dissolved faces of statues and trees bare of leaves.
Acid rain is formed when sulfur dioxide and nitrogen oxides released into the atmosphere react with oxygen, water and other chemicals to form sulfuric and nitric acids, which can dissolve in water vapor. Any rain (or snow, fog or even hail) that then falls has a more acidic pH than normal. That rain not only eats away at human stonework but can also harm animals and ecosystems, especially aquatic ones. And because these chemicals can be carried for hundreds of miles, pollution in one state or country can cause damage in another downwind.
Though sulfur dioxide and nitrogen oxides can come from natural sources, such as volcanoes, the majority comes from burning fossil fuels.
And once again, public alarm helped drive action. In the U.S., updates to the Clean Air Act created a cap-and-trade system for power plants—which at the time were largely coal-burning—that gradually ratcheted down the amount of sulfur dioxide and nitrogen oxides they could release, incentivizing the addition of pollutant-capturing scrubbers to smokestacks.
These pollutants are of growing concern in places such as India, where coal-fired power is generally increasing. But in the U.S., sulfur dioxide emissions have declined by 95 percent and nitrogen oxides by 89 percent, according to the Environmental Protection Agency. And wet sulfate deposition, a common indicator of acid rain, dropped by more than 70 percent between 1989–1991 and 2020–2022.

Beachgoers are sprayed with DDT as a new machine for distributing the insecticide is tested for the first time in 1945.
Bettmann/Getty Images
DDT
With publication of her book Silent Spring in 1962, biologist Rachel Carson helped ignite the modern environmental movement. The book detailed the devastating effects to wildlife—especially birds—from the widespread use of the insecticide DDT.
READ MORE: How the Environment Has Changed since the First Earth Day 50 Years Ago
The chemical was developed in the early 1900s and came into wider use during World War II to prevent the spread of malaria and typhus. But, as Scientific American wrote in 1944, “DDT’s effectiveness now in war may well be overshadowed by its value, as yet unutilized, in peace,” and it soon became widely used on crops, lawns, in homes and on pets. Some 1.35 billion pounds were sprayed in the U.S. alone over about 30 years, according to the EPA.
Public sentiment began to turn as some insects became resistant and the toll on wildlife became clear. DDT is persistent in the environment and accumulates up the food chain. It caused numbers of several bird species, including bald eagles, to nosedive as the shells of their eggs were so thin that they broke or failed to hatch.
The nascent EPA banned DDT in 1972, and levels in the environment have dramatically declined, though it is still found in trace amounts in soils. DDT is classified as a “probable human carcinogen,” and there are lingering effects—the grandchildren of women exposed to high amounts have higher rates of obesity and earlier age of their first period. Today it is also banned by international treaty for all uses except to control the spread of malaria.
One particularly bright spot to this story is the recovery of many bird species after the chemical was banned: bald eagle populations grew from only 417 known nesting pairs in 1963 to more than 70,000 today.

A view of the Los Angeles cityscape and downtown skyline shrouded in smog in May 1984.
Ron Eisenberg/Michael Ochs Archives/Getty Images
Smog
From the mid-20th century onward, Los Angeles became the poster child for cities choking in brownish-gray smog (a portmanteau of “smoke” and “fog”). As Scientific American wrote in 1964, “there is a growing realization that smog, beyond being a vexatious nuisance, may indeed present hazards to health.”
Smog arises from pollutants released by the burning of fossil fuels. In this case, nitrogen oxides and what are called volatile organic chemicals react with sunlight to create ground-level ozone, the principal component of smog. Though ozone is protective in the stratosphere, it is an irritant to the throat and lungs and can damage lung function.
In 1971 the EPA enacted National Ambient Air Quality Standards (NAAQS) on six common pollutants, including ground-level ozone, and those were strengthened in 1997. Since 1980, ozone pollution in the U.S. has declined by 29 percent, according to the EPA.
Smog is still a problem in U.S. cities, including Los Angeles, and the EPA under the Trump administration has proposed changes to various rules that could increase smog levels. Smog is also a growing concern in countries such as India that have rapidly industrialized.

