In order to improve care for people with multiple sclerosis (MS), we need to understand more about their disease. The way we measure the impact of MS on daily life has remained relatively unchanged for decades and is heavily reliant on clinic visits that may only occur once or twice each year. Unfortunately, this paradigm fails to capture the subtle mental and physical changes, possibly reflecting MS disease worsening, that can occur between visits. With the emergence of digital health there is the potential for daily monitoring of disease activity and progression in MS using tools such as Floodlight, a smartphone-based digital assessment developed by Roche and Genentech. Floodlight could help people with MS and healthcare professionals to have a greater understanding of the disease, ultimately leading to improved patient care. Approaches such as Floodlight may kick-start the use of novel measures to help detect whether symptoms of a disease are worsening. Floodlight and other digital tools aim to support Roche’s ability to develop medicines with smaller, more efficient, and more patientcentric trials.
THE UNPREDICTABILITY OF LIVING WITH MS
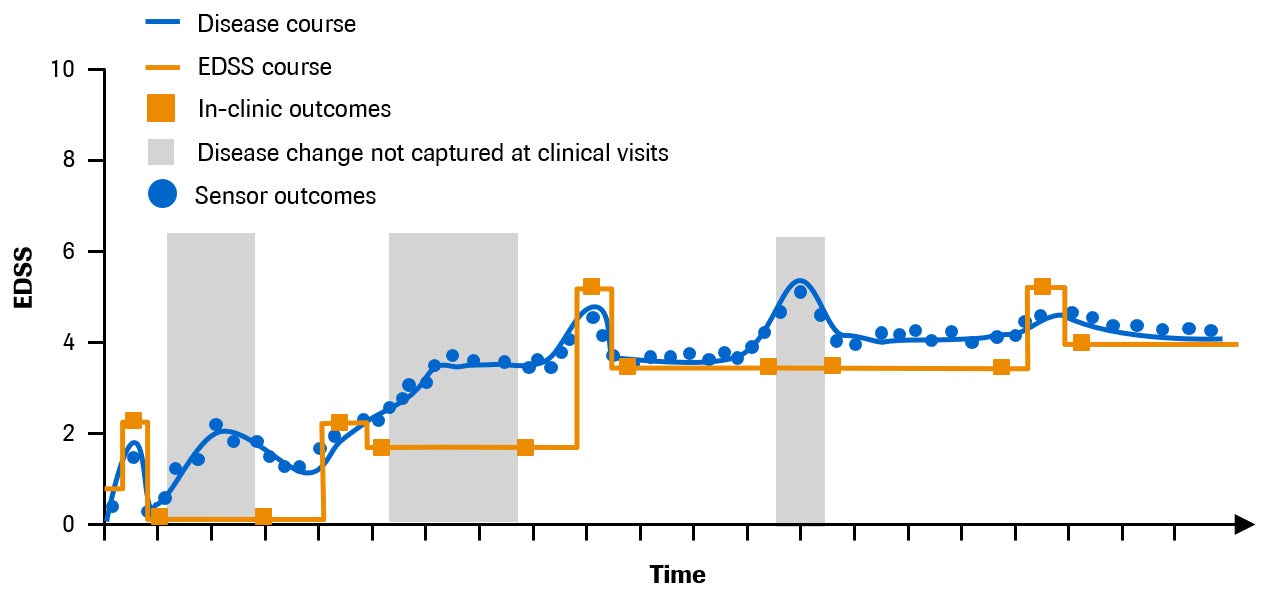
MS is an incurable chronic disease in which the body’s own immune system destroys tissue in the brain and spinal cord1. It affects 2.3 million people worldwide and is the leading cause of non-traumatic disability in young adults2. Symptoms may include visual disturbances, fatigue, pain, mobility problems and mental decline, although the specific disease course of MS is unpredictable and highly variable across individuals3 (Fig. 1). Many people with MS experience permanent disability.
The negative consequences of MS can be life-changing. Notably, MS is primarily diagnosed in active young adults in their twenties and thirties3, two-thirds of whom are women4. Put another way, MS has a substantial impact on people who have their whole lives ahead of them, who may be early on in their careers and making decisions about starting and raising families1.
day a person’s disease progression may be continuously monitored remotely7.
CHALLENGES FACING THE CURRENT STANDARDS OF CARE IN MS
The main goal of a healthcare provider involved in treating a person with MS is to manage disease activity and reduce the risk of disability worsening in a way that is meaningful to that individual. A critical factor for achieving this goal is the ability to monitor MS symptoms and disability progression, which is difficult with the current standards of care: clinic visits may be infrequent, appointment times tend to be short, and examinations can vary between clinicians. Consequently, the more subtle changes in disease progression that may occur between visits are missed (Fig. 1). Moreover, the tools typically used to assess symptoms are often irrelevant to an individual’s everyday life or impractical for routine assessment. For example, the Expanded Disability Status Scale (EDSS), which is a globally recognized assessment of disability in people with MS both in trials and clinical practice, is heavily focused on ambulation5 and may not adequately capture many other important symptoms for patients, such as upper limb function and cognition. Another commonly used measure, the nine-hole peg test, assesses hand function by measuring how quickly a person can place into and then remove nine pegs from nine holes, one at a time6. Measures such as these do not directly assess tasks that might be considered important to people with MS, such as writing or ability to work, making it difficult to translate findings into meaningful care.
The consequence of these challenges is delayed recognition of disease activity by the healthcare professional; a delay can subsequently impact timely treatment decisions and, ultimately, lead to irreversible disability. Real-time digital monitoring of MS outside of the clinic has the potential to overcome such challenges. Efforts are already ongoing to develop tools for enhanced monitoring of MS, and with the evolution of both mobile technology7 and wearable biosensors8, digital health tools promise change.
THE RISE OF NEW TECHNOLOGY
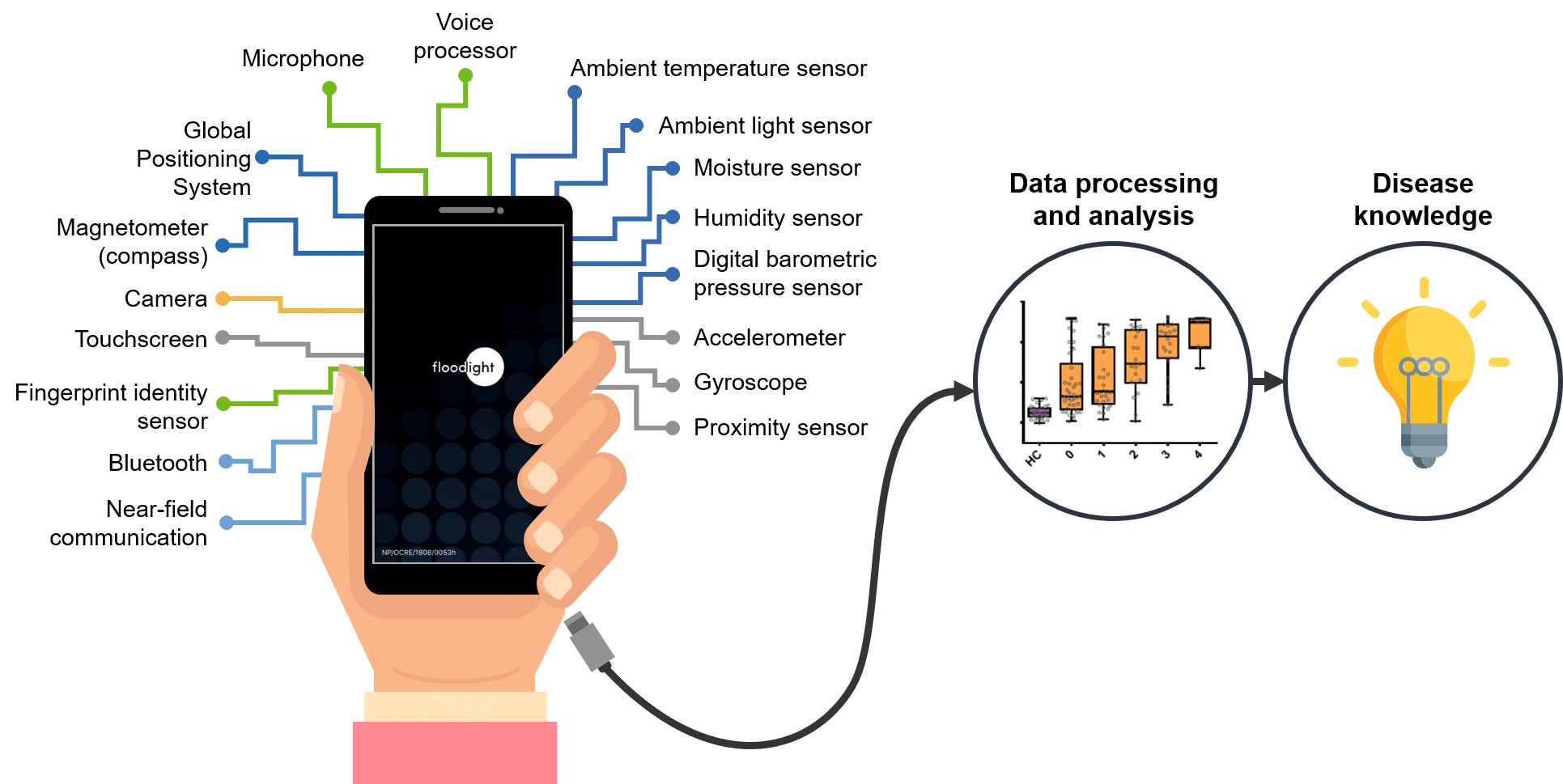
The rise of smartphones has given us the ability to connect with our family, friends and doctors, and to continuously monitor our health no matter where we are, every day.
Smartphones combine multiple, high-quality sensors into one indispensable device (Fig. 2). These sensors allow us to take basic measurements such as acceleration, and calculate parameters that describe how a person walks or the number of steps that they take during a day. Most smartphones can also sense geographic position, ambient light, voice and touchscreen pressure, and some even monitor atmospheric pressure. Utilizing sensors can turn a smartphone into a useful tool for monitoring heart rates or detecting falls7.
SMARTPHONE-BASED SELF-MONITORING OF MULTIPLE SCLEROSIS USING FLOODLIGHT
Floodlight is an app developed by Roche that contains a range of active and passive remote monitoring tests that measure a person’s ability to perform simple tasks on their smartphone (Fig. 3). The aim of Floodlight is to gain an understanding of the effects of MS on the brain, hands and body, and allow the symptoms of MS to be continuously monitored every day.
.jpg)
assessments, referred to as ‘active tests’.
REDISCOVERING MS WITH FLOODLIGHT
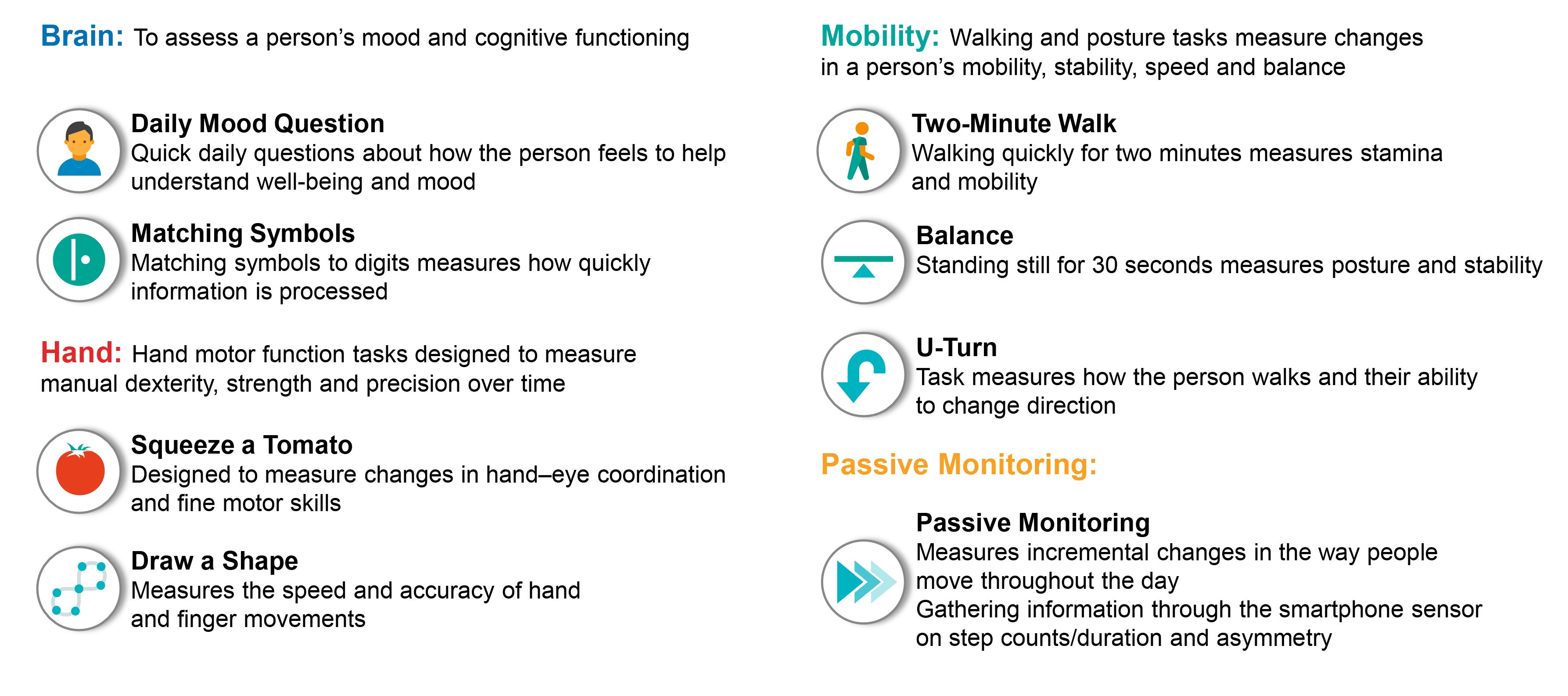
The Floodlight app consists of several tasks that measure cognition, hand motor function and mobility in order to collect precise, real-time data and that one day may track subtle changes in symptoms between doctor visits. Simple tasks were designed by physicians to help people with MS understand their personal journey. The tests are intended to enhance the findings from several gold standard in-clinic tests, accurately tracking disease activity and progression while at the same time providing a clearer and more continuous picture of the reality experienced by people living with MS (Fig. 4).
Consider hand motor function, which, as previously described, is typically measured by the nine-hole peg test. In the Floodlight app, one of the tests used to assess hand motor function is a test called draw a shape, in which users hold the smartphone in one hand and draw six different shapes of increasing difficulty on the touchscreen with the finger of the opposite hand9. The complete interaction with the touchscreen is recorded, performance features such as accuracy and speed are measured, and changes are tracked over time. The hope is that the draw a shape test is faster and more accurate, can be carried out every day, and reflects a person’s ability to write, a task that is particularly meaningful to people with MS.
EMPOWERING PEOPLE LIVING WITH MS
Giving people living with MS the opportunity to actively engage in understanding and managing their disease can be empowering. This is becoming increasingly important because therapeutic decisions are mostly made in partnership between the doctor and the patient, and there is evidence to suggest that involvement of people with MS in shared decision-making is central to improving their treatment satisfaction and adherence10, 11.
The hope is that one day, tools such as the Floodlight app will allow people with MS to do this by tracking their own disease journey on their smartphone, enabling them to accurately identify changes in their symptoms and to engage in informed patient-led discussions. Digital monitoring is not about replacing a healthcare professional. Rather, it has the potential to bring people together, make healthcare more personalized and improve the patient–clinician relationship12. In a recent exploratory study, we found that people living with MS who completed the Floodlight tests regularly over a six-month period were highly engaged and satisfied with the app, supporting the feasibility of using smartphonebased monitoring of MS in a person’s daily life.
MAKING DIGITAL HEALTHCARE A REALITY
Roche’s commitment to neuroscience extends beyond the development of new medicines. In the age of the digital healthcare revolution, tools like Floodlight represent a promising avenue to enable minimally intrusive, precise and continuous assessment of MS disease activity integrated into people’s daily routines. Implementing digital monitoring could identify disease progression earlier, leading to more rapid and appropriate treatment decisions, and therefore has the potential to improve long-term health in people with MS. With the advent of the digital healthcare revolution and the rapid advances in technology, it may be possible that one day, a patient’s disease can be passively monitored 365 days a year, using the technology that exists around us. To make the idea of digital healthcare a reality and ensure that we measure what matters to patients, collaborations are required between digital experts, healthcare providers, people with MS and the wider community of MS researchers. The digital assessments and tests need to be designed alongside people with MS, and the results need to be reported in a way that facilitates smooth integration into research and healthcare workflows. A cross-industry and ultimately even cross-healthcare system approach is required to define the standards of digital monitoring, with the ultimate goal of improving the lives of people living with MS.
REFERENCES
- Giovannoni, G. et al. Brain health: Time matters in multiple sclerosis. Mult. Scler. Relat. Disord. 9, Suppl. 1 S5–S48 (2016).
- Tillery, E. E., Clements, J. N. & Howard, Z. What’s new in multiple sclerosis? Ment. Health Clin. 7, 213–220 (2018).
- Compston, A. & Coles, A. Multiple sclerosis. The Lancet 359, 1221–1231 (2002).
- Multiple Sclerosis International Federation. Atlas of MS 2013: Mapping mutliple sclerosis around the world. 2013.
- Cadavid, D. et al. The EDSS-plus, an improved endpoint for disability progression in secondary progressive multiple sclerosis. Mult. Scler. 23, 94–105 (2017).
- Feys, P. et al. The nine-hole peg test as a manual dexterity performance measure for multiple sclerosis. Mult. Scler. 23, 711–720 (2017).
- Sim I. Mobile devices and health. N. Engl. J. Med. 381, 956–968 (2019).
- Bradshaw, M. J., Farrow, S., Motl, R. W. & Chitnis, T. Wearable biosensors to monitor disability in multiple sclerosis. Neurol. Clin. Pract. 7, 354–362 (2017).
- Midaglia, L. et al. Adherence and satisfaction of smartphone- and smartwatch-based remote active testing and passive monitoring in people with multiple sclerosis: Nonrandomized interventional feasibility study. J. Med. Internet Res. 21, e14863 (2019).
- Ben-Zacharia, A. et al. Impact of shared decision making on diseasemodifying drug adherence in multiple sclerosis. Int. J. MS Care 20, 287–297 (2018).
- Barbosa, C. D., Balp, M. M., Kulich, K., Germain, N. & Rofail, D. A literature review to explore the link between treatment satisfaction and adherence, compliance, and persistence. Patient Prefer. Adherence 6, 39–48 (2012).
- Warraich, H. J., Califf, R. M. & Krumholz, H. M. The digital transformation of medicine can revitalize the patient-clinician relationship. NPJ Digit. Med. 1, 49. eCollection (2018).
Writing and editorial assistance was provided by Eleanor Foy of Articulate Science and Liz LaFlamme of Health Interactions, funded by F. Hoffmann- La Roche Ltd.
AUTHORS
Mike Baker, Global Head Digital Health
F. Hoffmann-La Roche Ltd, Building 1, 14th Floor, Grenzacherstrasse 124, 4070 Basel, Switzerland
Johan van Beek, Group International Scientific Director
F. Hoffmann-La Roche Ltd, Building 1, 12th Floor, Grenzacherstrasse 124, 4070 Basel, Switzerland
Christian Gossens, Global Area Head Digital Biomarkers
F. Hoffmann-La Roche Ltd, Building 92, 7th Floor, Grenzacherstrasse 124, 4070 Basel, Switzerland

