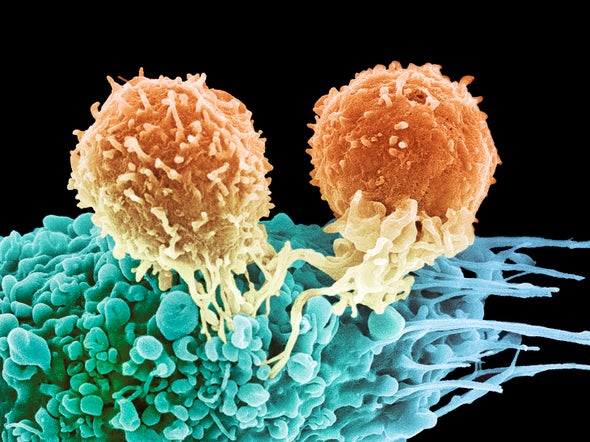
In 1891, William B. Coley—the head of the bone tumor service at Memorial Hospital in New York City—noticed an interesting phenomenon. Among certain cancer patients, the disease seemed to regress with the acquisition of a bacterial infection—not an uncommon occurrence in 19th century hospitals.
Curious, Coley began injecting patients with bacteria, and indeed, certain among them got better. Presumably, the injections set off an immune response that battled the bacteria and, subsequently, the cancer. But at the time, no one really understood the mechanism and the treatment was quickly abandoned with the advent of radiation and chemotherapy.
Today, this approach of harnessing the immune system to help fight cancer, known as Immuno-Oncology (I-O), is one of the fastest-growing areas in cancer research. Since the first agents became available more than seven years ago, the number of I-O treatments has expanded to include more than 26 therapies, with hundreds more under investigation. The troubling paradox of I-O is that while some patients benefit from treatment, too frequently I-O therapy doesn’t work at all, or the tumor learns to elude it over time.
This incongruity, referred to as I-O resistance, has emerged as one of the most important challenges for cancer researchers. There is an urgent need to better understand the mechanisms of resistance so we can make concerted efforts to overcome it.
Understanding the Science
There are two different types of I-O resistance. If an I-O therapy never works for a patient, that’s called primary resistance. In those cases, some characteristic of the tumor prevents the drug from working. Acquired resistance means that a treatment works at first and then stops after a period of time.
There are a number of ways tumors can manipulate the immune system to evade detection, such as interfering with signaling pathways that activate the immune response. “Tumors use the body’s own immunosuppressive mechanisms to send confusing signals to the immune system,” explains John Wherry, director of the institute for immunology and chair of the department of systems pharmacology and translational therapeutics at the University of Pennsylvania. “Cancer tells the immune system there’s something foreign, but tumors send signals telling the immune system to shut off and not damage tissue.”
To make matters more complicated, tumors can also use different immune escape mechanisms over the course of the disease. “It’s almost like a game of ‘whack-a-mole,’” said Wherry. “I-O therapy will often address one of these pathways, but then another springs up, bringing us back to square one.”
In addition to immune suppression pathways, resistance can also be influenced by tumor types. “In broad strokes, we can characterize tumors in two ways—immunologically ‘hot’ or ‘cold’. Hot, or inflamed, tumors typically have a pre-existing immune response and a high volume of mutations, which allow the immune system to better recognize the tumor,” said Michael Quigley, executive director of immuno-oncology resistance biology at Bristol-Myers Squibb. “In some cases, certain tumors have more mutations that make them immunologically hot and more likely to respond to I-O therapy.”
But as Quigley explains, cold tumors have a much less optimistic outlook. “These tumors can either have a low rate of mutations, which means they’re less likely to be registered as foreign invaders in the body and instead go unnoticed by the immune system, or use other means to subvert an immune response. In these situations, tumors almost never respond to currently available I-O therapies.”
.jpg)
Fighting the Resistance
The field of I-O research has expanded, but unfortunately, as the number of patients exposed to immunotherapies increases, so does the number of patients who become resistant. As a pioneer in the I-O field, Bristol-Myers Squibb saw first-hand the impact of I-O resistance and decided to tackle the issue head on.
The company established a dedicated I-O resistance team comprised of discovery and translational researchers from across the country working together to understand resistance mechanisms and uncover why some patients do not initially respond or stop responding to treatment.
To help researchers accomplish this lofty goal, Bristol-Myers Squibb invested in two state-of-the-art laboratories with an emphasis on resistance. The Redwood City, CA location, which opened in 2012, is home to the company’s I-O antibody discovery engine. The Cambridge, MA site is Bristol-Myers’ newest R&D facility, which includes a focus on identifying new targets and pathways for tumors that are refractory to current treatments.
“We realized early on that I-O resistance would require us to push the boundaries of science, and there’s no better place to do that than in the vibrant ecosystems of Cambridge and Redwood City,” said Saurabh Saha, SVP, global head of translational medicine at Bristol-Myers Squibb and lead of the Cambridge site. “Working together with our academic and industry collaborators just a stone’s throw away, we have pharma resources moving at the speed of a biotech with a laser focus on solving this critical challenge.”
Scientists are continuing to explore new avenues to understand patient responses, or lack thereof, to treatment. For instance, researchers are analyzing hundreds of consented samples to identify potential indicators—or biomarkers—of response, that can be used to predict which patients will be the best candidates for certain therapies.
Basket trials—clinical trials that evaluate one drug in a multitude of tumor types at the same time—are another weapon in the arsenal, allowing researchers to look back and analyze multiple mechanisms in a single study.
“At a patient level, we’re starting to be able to look retrospectively at who responded,” said Quigley. From that, scientists hope to find clues to help assign patients with the best treatment regimen from the start.
The Road Ahead
I-O resistance may prove to be one of oncology’s toughest challenges, but the outlook is hopeful. In fact, researchers have already started to identify approaches that may spur the development of new treatments.
For instance, Bristol-Myers Squibb researchers have hypothesized that patients with elevated levels of lymphocyte-activation gene-3 (LAG-3)—an immune checkpoint receptor protein—may benefit from strategies directed at the PD-1 and LAG-3 pathways. “We still have a lot of work to do, and it would by no means solve the entire problem, but this has potential for a segment of the population with a certain disease biology,” said Quigley.
While the fight against I-O resistance continues to move forward with speed, Quigley is quick to point out the ultimate purpose behind these efforts.
“Having lost my mother to cancer, I can tell you this is more than a scientific endeavor, it’s about preserving lives. While resistance has presented a significant hurdle, we are working urgently to unlock the power of the immune system and help as many cancer patients as possible live long, healthy lives,” said Quigley. “I’m immensely grateful to those who participate in clinical trials and donate samples to help advance our research. The opportunity to help others, people like my mother, is what brings us to work every day, and we won’t stop pushing forward until we uncover a way to help more patients with cancer.”