One of the great challenges when developing biologically active compounds is to produce ones that are nearly identical to those in nature–but not exactly. By swapping an atom here or there, synthetic chemists can more accurately probe the processes of life and discover what happens when they malfunction in various disorders. The same molecules could also be used to develop new drugs.
Problem: Such atomic-scale manipulation is difficult. Organic compounds are typically complex, and conventional ways of making them involve multi-step procedures. Those are time consuming and wasteful, since chemists have to stop and purify the product after each reaction has run its course.
Promising solution: In the past decade, so-called catalytic cascade reactions have gained favor as a way to circumvent these challenges. Just as a falling domino will cause its neighbor to topple, each reaction in a catalytic cascade leads naturally to the next one, minimizing the steps required. Dubbed one-pot reactions because they can be performed in a single container, cascade reactions are far faster than conventional ones.
Study: In a recently published paper in Nature Communications, Fei Ye, Matthias Beller and three colleagues, all at the Leibniz Institute for Catalysis in Germany, describe how they used catalytic cascade reactions to develop two new strategies to produce spiro-fused heterocycles. These organic molecules have been found to play key roles in biological processes, namely cellular metabolism, making them promising for developing new drugs.
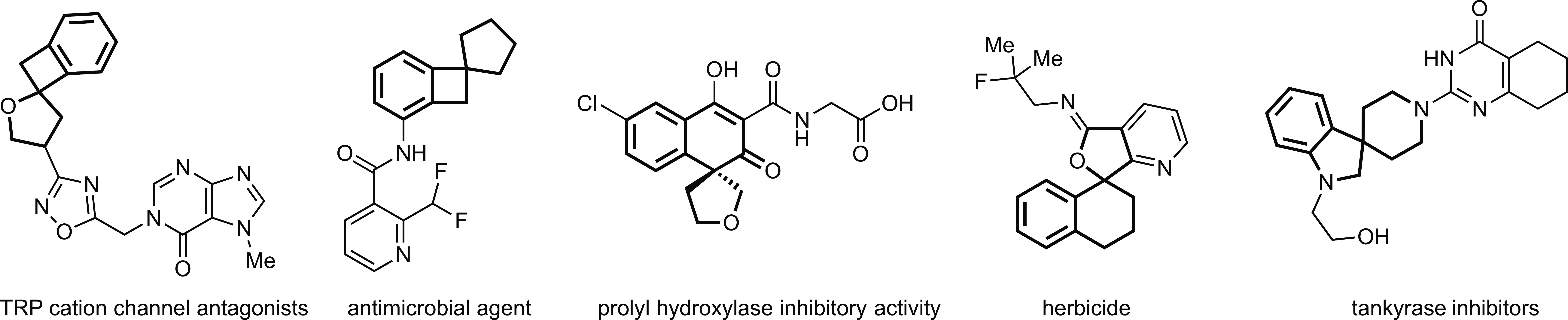
Main achievement: The team developed a benign and efficient catalytic process for biologically active compounds, which are otherwise difficult to produce. Specifically, they created fluorinated building blocks using a catalytic cascade reaction and two other key components–tailored palladium catalysts and allyl ammonium salts. This combination allowed the team to make diverse spiro-fused heterocycles rapidly and efficiently, without the need to purify intermediate compounds.
The team used their method to produce 48 different spiro-fused heterocycles. Some of these compounds were closely related to naturally occurring compounds except they had fluorine atoms instead of hydrogen ones at specific positions. These variants generally behave like their natural counterparts, but are more difficult for the body to metabolize.
Significance: This synthetic method could benefit other researchers investigating biological processes and metabolic disorders, and the results demonstrate the viability of the team’s process. Further valuable compounds could soon be on the way.
To read more about the research, explore the paper in Nature Communications.
 Fei Ye obtained his PhD from the University of Paris VI (Paris, France) in 2017. Since October 2018, he has worked as a postdoctoral fellow in the group of Prof. Dr. Matthias Beller at Leibniz Institute for Catalysis in Rostock, Germany. He is now an assistant professor at Hangzhou Normal University in Hangzhou, PR China, where he focuses on ligand synthesis, asymmetric catalysis and organofluorine chemistry.
Fei Ye obtained his PhD from the University of Paris VI (Paris, France) in 2017. Since October 2018, he has worked as a postdoctoral fellow in the group of Prof. Dr. Matthias Beller at Leibniz Institute for Catalysis in Rostock, Germany. He is now an assistant professor at Hangzhou Normal University in Hangzhou, PR China, where he focuses on ligand synthesis, asymmetric catalysis and organofluorine chemistry.


