Abbott is a global healthcare company that creates life-changing products—in diagnostics, medical devices, nutrition and branded generic pharmaceuticals—that help people and their families lead healthier lives. Abbott aims to provide accurate diagnostic tools that enable smarter, faster decisions and transform the way healthcare providers are managing patients. The company’s diagnostic technology spans the world of healthcare operations—with medical diagnostic instruments, tests, automation and informatics solutions for hospitals, reference labs, blood centers, emergency departments, physician offices and clinics. In line with this commitment, Abbott is striving to develop products to reduce cardiovascular disease (CVD), which is the number one cause of mortality worldwide. To address this disease burden, Abbott developed a cardiac-specific biomarker, high-sensitivity cardiac troponin I (hs-cTnI), to help doctors more accurately predict the chances of a heart attack or other cardiac event among adults who otherwise appear healthy.
CVD burden
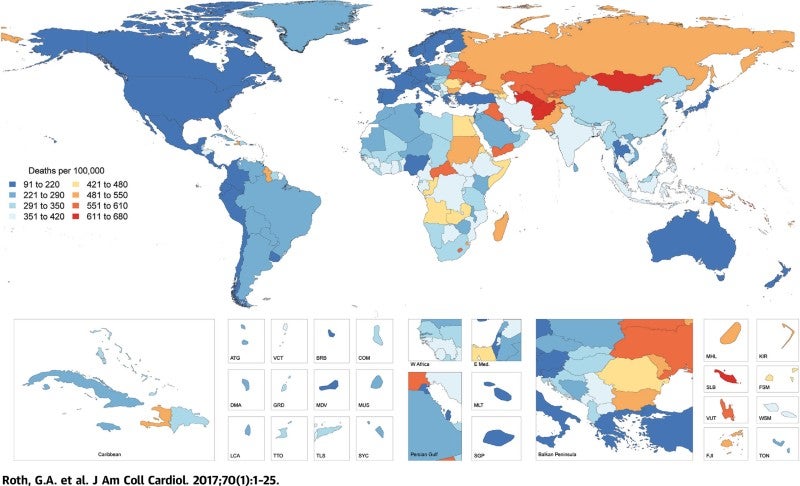
CVD has been the leading cause of mortality worldwide for the past 20 years and the number of deaths reached 8.9 million in 2019. According to the World Health Organization (WHO), ischemic heart disease is responsible for a substantial number of deaths at the global level, accounting for about 16% of the total. It is the leading cause of death in high-income and middle-income countries, and the third leading cause of death in low-income countries1. Overall, the prevalence of CVD increased from 271 million in 1990 to 523 million in 2019, and the CVD burden has continued to increase each year. Figure 1 depicts the global age-standardized mortality rate due to CVD in 2015, and it is projected that 31.7% of deaths worldwide will be attributable to CVD by 2030. In the past 20 years, there have been major improvements in awareness, assessment of classic risk factors and treatments such as statins. However, data suggest that in recent years there have been increasing trends in classical cardiovascular (CV) risk factors such as hypertension and obesity2. We intend to explore the limitations of current methods as well as novel approaches to curb this disease burden.
Classical risk factors and diagnostic tests
The terms CV risk factors and risk markers were conceptualized formally in the 1960s when the findings of the Framingham study started to appear in the literature. Based on the epidemiological definition, a risk marker is a feature or characteristic that appears early in a human’s life and can predict future disease. In CVD, some risk factors are fixed and cannot be altered, such as a family history of premature coronary heart disease, whereas others such as acquired behaviours like smoking can be modified—meaning that the cessation of smoking can reduce the risk of future CVD. Established risk factors are hyperlipidaemia and elevated low-density lipoprotein (LDL), smoking status, diabetes and obesity. Global prediction models using some of the established CV risk factors have improved the detection of disease risk; however, it seems that 20% of all events occur in their absence. To account for the residual CV risk, several other novel risk markers or diagnostic tests have been developed with different pathophysiological pathways; these include some representing inflammation (such as high-sensitivity C-reactive protein or hs-CRP), altered thrombosis (such as homocysteine), oxidative stress (such as oxidized LDL) or altered lipids (such as lipoprotein(a)).
In practical terms, a family history of premature coronary artery disease and obesity are among the most common CV risk factors taken into consideration when reviewing a subject. We also determine the underlying issues and comorbidities, which may include hypertension, dyslipidaemia and diabetes, with routine diagnostic tests. One of the most common of these is the lipid profile, whereby total cholesterol, LDL and high-density lipoprotein (HDL) are reviewed as indicators and even as treatment targets for statin therapy. Such common factors are utilized in various combinations when calculating the overall CVD risk. Further work-up of the subject based on clinical suspicion and risk category may include cardiac imaging studies. However, these are less commonly used and are limited by their cost and availability.
Current risk-prediction tools
Traditional CV risk prediction does not identify everyone who will develop CVD, with up to 50% of individuals having none or only one risk factor at the time of diagnosis3. Although traditional risk estimations perform moderately well, there remain significant limitations to their use in the prevention of CVD; this is especially relevant at an individual level, where the clinician needs not only to correctly identify those at increased risk, but also to weigh up the importance of each risk factor and determine who needs medical therapy in addition to lifestyle advice.
Many risk-estimation systems are based on a core set of CV risk factors, and participants are selected randomly either from the general population4 or from those presenting to their general practitioner5. All these risk-scoring systems show a good level of discrimination for CV events, with the area under the receiving operator curve ranging from 0.73 to 0.82 (ref. 6). However, adopting these risk-scoring systems to guide current clinical practice has limitations. First, most of the scoring systems, except QRISK1 and QRISK2, have been developed from old prospective cohorts with participants recruited during the 1980s and 1990s. Second, applying risk-estimation scores to regions with different baseline rates of CVD will lead to either under- or over-estimation of risk as a result of miscalibration6. Third, incorporating new risk factors including biomarkers such as hs-CRP has been disappointing in improving discrimination, with age and gender alone contributing to 0.70 of the area under the receiver operating curve statistic. None of the risk-estimation scores to date incorporate a direct measure of cardiac injury such as cardiac troponins.
High-sensitivity troponins
Cardiac troponins are structural proteins that can be found mainly in the myofibrils of cardiomyocytes. The cells of the myocardium contain three types of troponin: troponin I (TnI), troponin T (TnT) and troponin C. Troponin C is also found in skeletal muscle, whereas TnI and TnT are specific to cardiac myocytes and are therefore clinically useful for measuring damage to the myocardium. According to the International Federation of Clinical Chemistry and Laboratory Medicine, one key characteristic of a hs-cTn assay is the ability to detect troponin in ≥50% of healthy individuals7. This is made possible by the additional requirement for improved precision of the test, thus lowering the limit-of-detection threshold. This has expanded the potential use of cardiac troponins from diagnostic tools in the setting of acute coronary syndrome in conjunction with clinical findings to biomarkers for risk stratification in individuals without known CVD.
For a biomarker to have clinical utility as a screening tool in the general population, it must meet some specific criteria. hs-cTnI is a good candidate for CV risk assessment of the general population for several reasons, including the following8. First, cardiac specificity, as high-sensitivity assays are specific to cardiac troponins and can detect low levels in a substantial number of individuals in the general population. Second, plausibility, as high-sensitivity troponins are a known marker of myocardial injury. Third, temporality and predictive value, as hs-cTnI predicts incident CV events in the general population. Fourth, consistency, as multiple observational studies have demonstrated the association between baseline hs-cTnI and CV outcomes across different situations (for example, different populations and origins). Fifth, dose-response gradient, as higher troponin levels are associated with higher CV risk. Sixth, risk modification, as hs-cTnI reduction parallels CV risk reduction. Seventh, response to interventions such as statin initiation, which reduces hs-cTnI levels. Eighth, additive value, as hs-cTnI performs more accurately when added to established CV risk-prediction tools such as Systematic COronary Risk Evaluation (SCORE) and the Framingham score. Ninth, cost-effectiveness, as assessing CVD risk with hs-TnI in the general population and treating those at high risk with statins is a cost-effective strategy8.Moreover, cardiac TnI measured at low levels can be detected in individuals with common cardiac conditions and CV risk factors, which implies that it can reveal subclinical myocardial injury.
hs-cTns in the general population
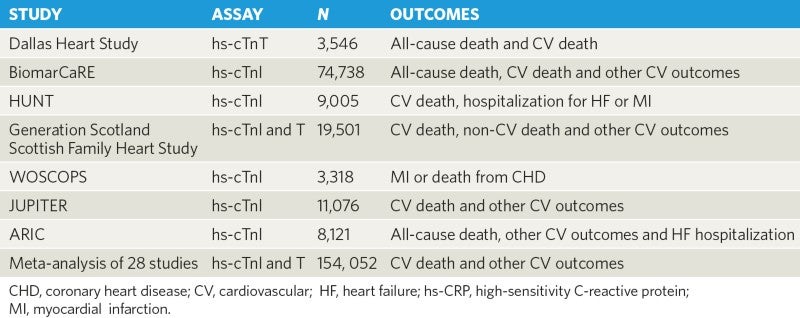
hs-cTns are predictors of adverse CV events in the general population9. A recent study-level systematic review and meta-analysis reported that hs-cTn concentrations within the normal reference range were associated with an increased risk of CV outcomes in the general population10. Table 1 summarizes the main studies of high-sensitivity troponins. The evidence is robust that hs-cTnI in particular can predict the risk of incident CV events such as mortality, myocardial infarction and heart-failure hospitalization.
High-sensitivity troponins and treatments
The latest generation hs-cTnI assay was evaluated in the West of Scotland Coronary Prevention Study (WOSCOPS)11, which was a seminal and influential randomized controlled trial of statin therapy in the primary prevention of coronary heart disease. In multivariate models, baseline troponin concentrations remained an independent marker of future CV risk, with more conventional risk factors such as LDL cholesterol failing to demonstrate associations. It was shown that those individuals with troponin concentrations ≥5 ng/L had a three-fold greater absolute risk reduction on pravastatin compared to those <5 ng/L (ref. 11). This concentration was also the optimal threshold for risk stratification in the emergency department12. This was the first demonstration that troponin concentrations are reduced by statin therapy; the magnitude of this reduction at one year was an independent predictor of future coronary events and identified those who responded to therapeutic intervention. In contrast, there was no relationship between the reduction in coronary events on statin therapy and the extent of LDL cholesterol lowering11.
hs-cTns compared to cardiac imaging
Several studies have explored the association of high-sensitivity troponins and cardiac function as measured with imaging modalities. In healthy subjects screened with cardiac magnetic resonance imaging (MRI), hs-cTns were associated with cardiac morphology (that is, cardiac mass and volume). hs-cTnI was also independently associated with multidirectional strain, which is an index of cardiac function. This association with cardiac morphology and function might indicate that cardiac troponins can detect subclinical myocardial damage or injury in otherwise healthy individuals13. Coronary artery calcification as a marker of subclinical coronary atherosclerosis detected by computed tomography can provide prognostic information when added to classical CV risk factors. There was an association between coronary artery calcification—quantified by the Agatston score—and hs-cTnI in middle-aged subjects without previous CVD. More specifically, individuals in the highest quartile of hs-cTnI had 56% increased odds of having an Agatston score >0 and 82% increased odds of having an Agatston score ≥100 compared to those in the lowest quartile. This indicates that hs-cTnI can predict the burden of atherosclerosis in individuals without previous CVD. In another study, investigators enrolled 70-year-old volunteers and performed late-enhancement cardiac MRI at baseline and five years later. The purpose of this study was to investigate whether hs-cTnI can predict the development of new undiagnosed myocardial infarction. Baseline concentrations were associated with new or larger undiagnosed myocardial infarctions14. These findings imply that troponins are associated with early signs of subclinical myocardial injury and atherosclerosis, and might potentially help provide early signs of underlying coronary artery disease in asymptomatic individuals.
hs-cTn implementation
While high-sensitivity troponins have been utilized for several years in the acute setting to help manage patients with suspected myocardial infarction, they are less known for their capability to prevent CVD. For this paradigm shift in thinking to occur, it will be crucial to generate awareness. Additionally, there is a question as to how to implement a tool that is so engrained in clinical thinking as an emergency biomarker and not a risk-management biomarker. One suggestion is to review where hs-cTns fit into current CV risk strategies (Table 2 and Figure 2).
.jpeg)
OUR APPROACH TO REDUCING CVD BURDEN: THE IMPLEMENTATION REGISTRY
A medical registry has been defined as “a database of identifiable persons containing a clearly defined set of health and demographic data collected for a specific public-health purpose”15. Medical registries are a valuable source of epidemiological data that can help with disease surveillance. Modern registries can also be used in healthcare systems and can inform physicians and healthcare policymakers, as they can identify healthcare gaps and unmet needs in disease management. In this context, a CV risk registry can be a valuable tool to inform physicians about baseline CV risk assessment using a cardiac biomarker.

Our implementation registry acts as a patient-care application and includes baseline CV risk assessment based on the high-sensitivity troponin levels; it ultimately leads to improvement of CV care through lifestyle changes (such as diet, smoking cessation and exercise recommendations), and treatment initiation or intensification (for example with lipid-lowering or blood-pressure medications), while at the same time evaluating individuals’ outcomes and actively monitoring the disease risk. This can be done in line with a health check or a wellness programme at a hospital or a chain of linked hospitals, or even in the context of a countrywide CV-screening programme. Several countries worldwide have initiated public health-screening programmes with the aim of reducing CV burden and mortality rates in the future. This registry approach blends into the standard of practice for CV risk assessment with the addition of hs-cTnI being the only exception. Clinicians react to hs-cTnI based on the concentration and the overall risk profile of the subject. They choose a course of action based on their clinical judgement along with the additional knowledge of hs-cTnI. At the baseline visit, there is a recommendation for follow-up visits as per the standard practice (that is, depending on the risk profile). Follow-up data are captured and include outcome data (on hospitalizations and CV mortality) as well as hs-cTnI measurements at the physician’s discretion. Sample biobanking during such registries can be used to help future research by exploring the utility of new and novel biomarkers (ClinicalTrials.gov NCT04903041).
Data from these implementation registries can be used to inform policymakers and physicians aiming to reduce the CV burden and manage their patients in a more efficient way. According to recent publications, assessing CVD risk with hs-TnI in a general population and subsequently referring those at high risk to take preventive measures would be cost-effective. Based on these findings, fewer than 200 subjects need to be screened to prevent one CVD event within 10 years16. Thus, these registries can be used as a benchmark to inform public-health strategic actions through screening programmes; act as a basis for future pragmatic cluster-randomized clinical trials aimed at evaluating the effectiveness of troponin-guided treatment both in primary and secondary prevention; and even help explore the value of simple troponin measurements as a screening strategy in the broader population.Abbott’s leadership in this area has driven research collaborations with leading healthcare institutions globally. These have focused on unique cardiac registries to aid in implementation, monitoring and improving the outcomes of patients who are at risk for CVD events, which will have a measurable outcome on disease burden in the future.
AUTHORS
Christos Varounis and Agim Beshiri
References
-
World Health Organization. Newsroom https://www.who.int/news-room/fact-sheets/detail/the-top-10-causes-of-death (2020).
-
Roth, G.A. et al. J. Am. Coll. Cardiol. 70, 1–25 (2017).
-
Khot, U.N. et al. JAMA 290, 898–904 (2003).
-
D’Agostino, R.B. Sr et al. Circulation 117, 743–753 (2008).
-
Hippisley-Cox, J. et al. Heart 94, 34–39 (2008).
-
Cooney, M.T. et al. J. Am. Coll. Cardiol. 54, 1209–1227 (2009).
-
Wu, A.H.B. et al. Clin. Chem. 64, 545–655 (2018).
-
Farmakis, D. et al. Eur. Heart. J. 41, 4050–4056 (2020).
-
Omland, T. et al. Clin. Chem. 61, 646–656 (2015).
-
Willeit, P. et al. J. Am. Coll. Cardiol. 70, 558–568 (2017).
-
Ford, I. et al. J. Am. Coll. Cardiol. 68, 2719–2728 (2016).
-
Shah, A.S. et al. Lancet 386, 2481–2488 (2015).
-
Aw, T.C. et al. Sci. Rep. 8, 15409 (2018).
-
Barbier, C.E. et al. Clin. Chem. 60, 1327–1335 (2014).
-
Solomon, D.J. et al. Public Health Rep.106, 142–150 (1991).
-
Jülicher, P. & Varounis, C. Eur. Heart J. Qual. Care Clin. Outcomes, qcab005 (2021).

