
What is Type 1 Diabetes? Here’s Your 5-Minute Primer
What happens when your body suddenly stops making the one hormone that keeps your blood sugar in check?

What is Type 1 Diabetes? Here’s Your 5-Minute Primer
What happens when your body suddenly stops making the one hormone that keeps your blood sugar in check?

Shimon Sakaguchi Reflects on How Hunting for a Mysterious T Cell Earned Him a Nobel Prize
Nobel laureate Shimon Sakaguchi reflects on what role of regulatory T cells have in peripheral immune tolerance and how the cells could transform treatment for cancer, autoimmune disease and organ transplant rejection


Nobel Prizes, COVID Vaccine Updates and Malnutrition in Gaza
The CDC updates COVID vaccine guidance and stirs controversy over childhood immunizations. And global health experts warn of rising child malnutrition in Gaza.

What It’s like to Be the President’s Doctor
A former White House physician reveals the medical realities of caring for the president of the U.S.
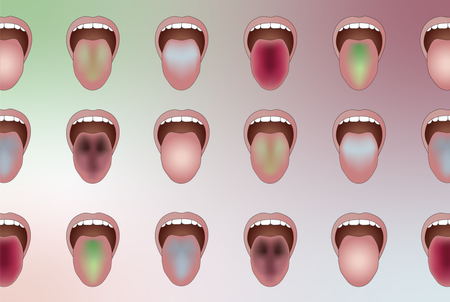
AI Reads Your Tongue Color to Reveal Hidden Diseases
Inspired by principles from traditional Chinese medicine, researchers used AI to analyze tongue color as a diagnostic tool—with more than 96 percent accuracy

2025 Chemistry Nobel Goes to Molecular Sponges That Purify Water, Store Energy and Clean Up the Environment
Three scientists, including one from the U.S., share the 2025 Nobel Prize in Chemistry for developing “metal-organic frameworks,” versatile molecular cages that can trap contaminants, store energy and possibly deliver drugs to specific areas of the body
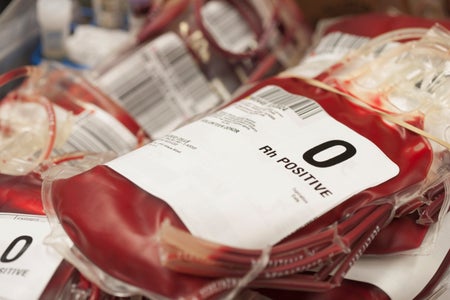
Scientists Perform First-of-Its-Kind Transplant Using Kidney with a Converted Blood Type
A man diagnosed with brain death received a kidney that was modified to be type O, which is compatible with all blood types

2025 Nobel Prize in Physiology or Medicine Awarded for Discoveries of How the Body Puts the Brakes on the Immune System
Mary E. Brunkow, Fred Ramsdell and Shimon Sakaguchi shared the Nobel prize for their work on peripheral immune tolerance, a process that is key to organ transplants and treatment of autoimmune diseases
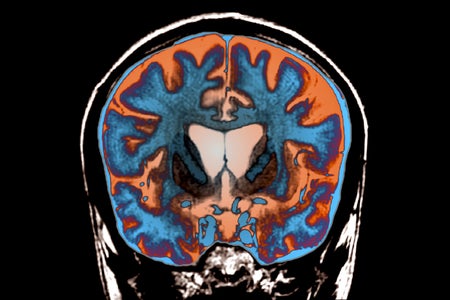
How Scientists Finally Found a Treatment That Slows Huntington’s Disease
After years of heartbreak, researchers have found an experimental treatment that can slow the progression of Huntington’s disease, according to early results from a small clinical trial

Is Tylenol Safe for Children?
President Trump says Tylenol is not safe for young children. Here’s what the science says about acetaminophen

Why It's Dangerous to Avoid Tylenol While Pregnant
Untreated fevers during pregnancy can cause more harm than taking acetaminophen will
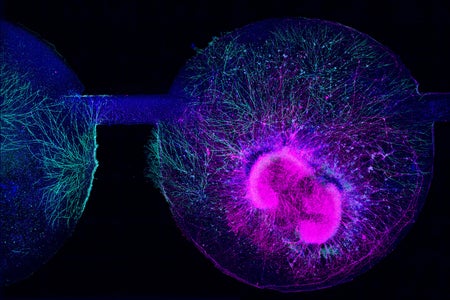
NIH Funds New Autism Studies on Genes and Environment as Trump Focuses on Tylenol
The National Institutes of Health is investing $50 million into research on genetic and environmental factors underlying autism—news that was eclipsed by President Donald Trump’s recent controversial claims about acetaminophen