Among the 10 leading causes of death, Alzheimer’s disease, which ranks as number six, is the only one that cannot be prevented, slowed or cured. While deaths from heart disease have decreased by 14% since 2000, deaths from Alzheimer’s disease (AD) have increased by 89% in that same timeframe, says the Alzheimer’s Association. But Nicola Corbett, a research associate at the Hooper lab at the University of Manchester in the U.K., is using induced pluripotent stem cells (iPSCs) to expose the cellular mechanisms behind AD. Consequently, she envisions a future where Alzheimer’s disease isn’t “such a huge, unresolved problem” and dreams of someday churning out research to help solve the AD jigsaw puzzle.
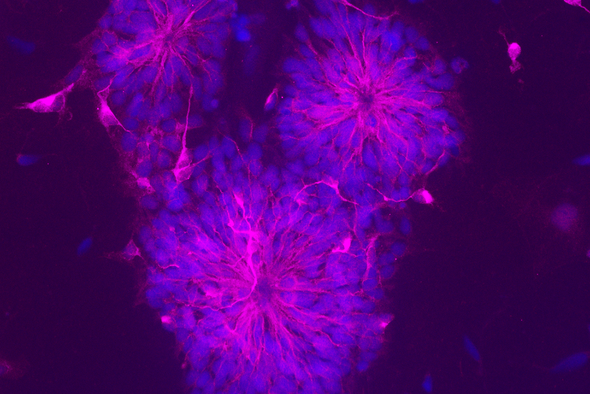
“We don’t really know where, or when, Alzheimer’s even starts in the brain, and I found that fascinating,” Corbett says. She’s currently studying the cellular biology of neurons that are derived from iPSCs, which were originally fibroblasts taken from Alzheimer’s disease patients and healthy aged patients. By comparing these neurons, Corbett hopes to uncover the differences that arise in AD. By using patient-derived iPSC neurons, she’s opening up the possibility of studying the pathophysiology of Alzheimer’s disease at the level of human cells.
Plenty of scientists have used iPSCs to make mature cells, including neurons, but Corbett aims to develop a cellular model of AD. Still, she admits that even the process of differentiating iPSCs into neurons is time-consuming and often times quite challenging — “some days, the cells just don’t want to play ball,” she says — but watching them turn into neurons is thrilling. “I always love looking down the microscope at them and finding new things,” she says.
Once she gets this process working regularly, she can turn to building and applying her model system. With a regular supply of neurons from AD patients, Corbett will next try to show that patient-derived iPSCs make a good model of the disease to use in testing novel strategies. If that works out, she can start to test potential treatments. This will mean looking for something that delays — or better yet, reverses — any differences between the diseased and healthy neurons.
Beyond battling such a widespread and debilitating disease, Corbett advocates for more women in STEM. To encourage tomorrow’s women scientists, Corbett keeps her passion for science alive and challenges stereotypes that can prevent young girls from pursuing a career in science. Putting this passion to work, Corbett hopes to help future generations of women scientists, as well as our aging population.
In thinking of the lack of medication for AD, Corbett says, “This is what neuroscientists, including myself, are working hard to change.”
This article is part of the Champions of Science series from Scientific American Custom Media and Johnson & Johnson Innovation.