On August 20, 2020, Russian opposition politician Alexei Navalny drank tea laced with a nerve agent at a Siberian airport and became gravely ill. Although he recovered after months of treatment, fast-acting nerve agents often overwhelm victims within hours, if not minutes.
Nerve agent attacks in espionage, civil wars, and terrorism are “a growing international concern,” according to a 2021 U.S. Congressional Research Service report. Current treatments can save someone from most nerve agents if applied quickly after exposure, but there is no safe pretreatment that offers extended protection. As a result, a victim who does not have immediate access to a nerve gas antidote will succumb to poisoning, sometimes within minutes.
Now U.S. Army scientists have developed a gene-based pretreatment that may one day offer long-term protection against a variety of these poisons.
Tackling toxicants
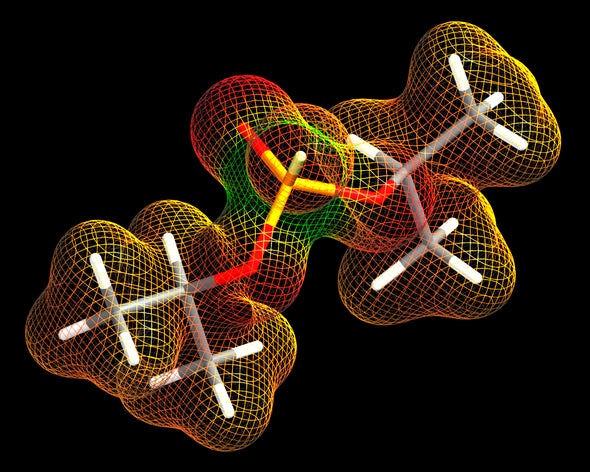
German scientists developed the first nerve agent, tabun, in 1936. Also known as GA, it was the first of a family of organophosphates that includes sarin (GB), soman (GD) and cyclosarin (GF). Absorbing, inhaling or eating these compounds, which are known collectively as G-type nerve agents, interferes with neurotransmitters and can cause death by cardiac or respiratory failure. Patients who survive may suffer long-lasting nerve and brain damage.
One pretreatment, pyridostigmine bromide, protects against soman if followed by rapid administration of other medications, but it causes other health problems, possibly even Gulf War Syndrome. So far, no safe and effective pretreatment exists for sarin or cyclosarin.
At the U.S. Army Medical Research Institute of Chemical Defense (USAMRICD) in Aberdeen, Maryland, researchers have been searching for enzymes that can destroy nerve agents before they cause damage. Butyrylcholinesterase, for example, breaks down G-type nerve agents in animal studies, but it takes one molecule of enzyme for every molecule of toxicant.
“You need a whopping amount of the enzyme,” says USAMRICD research chemist Nageswararao Chilukuri. Plus, this enzyme only lasts about a week in an animal’s blood.
Chilukuri wanted to find an enzyme that would destroy one toxic molecule after another, remain stable in blood, and not cause side effects.
DNA delivery
He found some of those features in a liver enzyme called paraoxonase 1, a single molecule of which can neutralize tens of thousands of organophosphate molecules. Using adenoviral vectors, Chilukuri inserted the human paraoxonase 1 gene into the DNA of mice. The animals’ livers produced paraoxonase 1 (PON1), but only for about one week.
At USAMRICD, Chilukuri was able to modify his approach, he says, because “the management tries to help you get what you want.” Backed by Army funding, Chilukuri switched to an adeno-associated virus vector that’s already approved for human gene therapy. Then he inserted the gene for a recombinant amalgam of rabbit, mouse and human paraoxonase 1 called PON1-IF11, which hydrolyzes nerve agents much faster.
“That changed the whole picture,” he says. “I was able to get very good yield of the enzyme, and I was able to make milligram levels of full-length enzymes.”
In mice, his team showed that PON1-IF11 provides months of protection against all four G-type nerve agents, they reported in Science Translational Medicine. G-type agents are similar enough chemically that an enzyme that breaks down one of them will break down all of them. The scientists plan to continue their work to prove that the gene therapy is safe for human use. Chilukuri is optimistic. Researchers at USAMRICD, he says, “are interested in achieving a common goal” of showing that this method works in humans.
To explore careers in the U.S. Army Medical Department (AMEDD), click here.